SARM1 Ablation Is Protective and Preserves Spatial Vision in an In Vivo Mouse Model of Retinal Ganglion Cell Degeneration
- PMID: 35163535
- PMCID: PMC8835928
- DOI: 10.3390/ijms23031606
SARM1 Ablation Is Protective and Preserves Spatial Vision in an In Vivo Mouse Model of Retinal Ganglion Cell Degeneration
Abstract
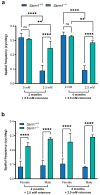
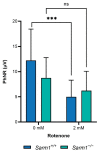
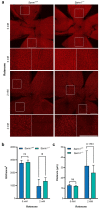
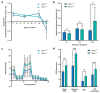
The challenge of developing gene therapies for genetic forms of blindness is heightened by the heterogeneity of these conditions. However, mechanistic commonalities indicate key pathways that may be targeted in a gene-independent approach. Mitochondrial dysfunction and axon degeneration are common features of many neurodegenerative conditions including retinal degenerations. Here we explore the neuroprotective effect afforded by the absence of sterile alpha and Toll/interleukin-1 receptor motif-containing 1 (SARM1), a prodegenerative NADase, in a rotenone-induced mouse model of retinal ganglion cell loss and visual dysfunction. Sarm1 knockout mice retain visual function after rotenone insult, displaying preservation of photopic negative response following rotenone treatment in addition to significantly higher optokinetic response measurements than wild type mice following rotenone. Protection of spatial vision is sustained over time in both sexes and is accompanied by increased RGC survival and additionally preservation of axonal density in optic nerves of Sarm1-/- mice insulted with rotenone. Primary fibroblasts extracted from Sarm1-/- mice demonstrate an increased oxygen consumption rate relative to those from wild type mice, with significantly higher basal, maximal and spare respiratory capacity. Collectively, our data indicate that Sarm1 ablation increases mitochondrial bioenergetics and confers histological and functional protection in vivo in the mouse retina against mitochondrial dysfunction, a hallmark of many neurodegenerative conditions including a variety of ocular disorders.
Keywords: NAD+; NADase; axon degeneration; retinal degeneration; sterile alpha and Toll/interleukin-1 receptor motif-containing 1 (SARM1).
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Role of SARM1 and DR6 in retinal ganglion cell axonal and somal degeneration following axonal injury.Exp Eye Res. 2018 Jun;171:54-61. doi: 10.1016/j.exer.2018.03.007. Epub 2018 Mar 8. Exp Eye Res. 2018. PMID: 29526794 Free PMC article.
-
SARM1 deficiency promotes rod and cone photoreceptor cell survival in a model of retinal degeneration.Life Sci Alliance. 2020 Apr 20;3(5):e201900618. doi: 10.26508/lsa.201900618. Print 2020 May. Life Sci Alliance. 2020. PMID: 32312889 Free PMC article.
-
Loss of Sarm1 reduces retinal ganglion cell loss in chronic glaucoma.Acta Neuropathol Commun. 2024 Feb 8;12(1):23. doi: 10.1186/s40478-024-01736-9. Acta Neuropathol Commun. 2024. PMID: 38331947 Free PMC article.
-
A Role for SARM1 in Photoreceptor Cell Death.Adv Exp Med Biol. 2025;1468:183-187. doi: 10.1007/978-3-031-76550-6_30. Adv Exp Med Biol. 2025. PMID: 39930193 Review.
-
The chemical biology of NAD+ regulation in axon degeneration.Curr Opin Chem Biol. 2022 Aug;69:102176. doi: 10.1016/j.cbpa.2022.102176. Epub 2022 Jul 1. Curr Opin Chem Biol. 2022. PMID: 35780654 Free PMC article. Review.
Cited by
-
Optimisation of AAV-NDI1 Significantly Enhances Its Therapeutic Value for Correcting Retinal Mitochondrial Dysfunction.Pharmaceutics. 2023 Jan 18;15(2):322. doi: 10.3390/pharmaceutics15020322. Pharmaceutics. 2023. PMID: 36839646 Free PMC article.
-
Rotenone-Induced Optic Nerve Damage and Retinal Ganglion Cell Loss in Rats.Biomolecules. 2024 Aug 23;14(9):1047. doi: 10.3390/biom14091047. Biomolecules. 2024. PMID: 39334813 Free PMC article.
-
Downregulation of SARM1 Protects Retinal Ganglion Cell Axonal and Somal Degeneration Via JNK Activation in a Glaucomatous Model of Ocular Hypertension.Invest Ophthalmol Vis Sci. 2024 Nov 4;65(13):7. doi: 10.1167/iovs.65.13.7. Invest Ophthalmol Vis Sci. 2024. PMID: 39499508 Free PMC article.
-
SARM1 is an essential component of neuronal Parthanatos.bioRxiv [Preprint]. 2025 May 15:2025.05.14.654090. doi: 10.1101/2025.05.14.654090. bioRxiv. 2025. PMID: 40463025 Free PMC article. Preprint.
-
SARM1: The Checkpoint of Axonal Degeneration in the Nervous System Disorders.Mol Neurobiol. 2025 Jul;62(7):9240-9257. doi: 10.1007/s12035-025-04835-3. Epub 2025 Mar 17. Mol Neurobiol. 2025. PMID: 40097763 Review.
References
MeSH terms
Substances
Grants and funding
- 16/IA/4452/SFI_/Science Foundation Ireland/Ireland
- 16/IA/4376/SFI_/Science Foundation Ireland/Ireland
- GOIPG/2017/1631/Irish Research Council
- HRAPOR-2015-1140/Health Research Board of Ireland
- MRCG-2012-4/Fighting Blindness Ireland - Health Research Board of Ireland - Health Research Charities Ireland
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

