Enhanced Bacteremia in Dextran Sulfate-Induced Colitis in Splenectomy Mice Correlates with Gut Dysbiosis and LPS Tolerance
- PMID: 35163596
- PMCID: PMC8836212
- DOI: 10.3390/ijms23031676
Enhanced Bacteremia in Dextran Sulfate-Induced Colitis in Splenectomy Mice Correlates with Gut Dysbiosis and LPS Tolerance
Abstract
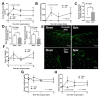
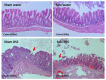
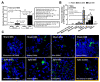
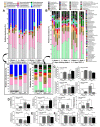
Because both endotoxemia and gut dysbiosis post-splenectomy might be associated with systemic infection, the susceptibility against infection was tested by dextran sulfate solution (DSS)-induced colitis and lipopolysaccharide (LPS) injection models in splenectomy mice with macrophage experiments. Here, splenectomy induced a gut barrier defect (FITC-dextran assay, endotoxemia, bacteria in mesenteric lymph nodes, and the loss of enterocyte tight junction) and gut dysbiosis (increased Proteobacteria by fecal microbiome analysis) without systemic inflammation (serum IL-6). In parallel, DSS induced more severe mucositis in splenectomy mice than sham-DSS mice, as indicated by mortality, stool consistency, gut barrier defect, serum cytokines, and blood bacterial burdens. The presence of green fluorescent-producing (GFP) E. coli in the spleen of sham-DSS mice after an oral gavage supported a crucial role of the spleen in the control of bacteria from gut translocation. Additionally, LPS administration in splenectomy mice induced lower serum cytokines (TNF-α and IL-6) than LPS-administered sham mice, perhaps due to LPS tolerance from pre-existing post-splenectomy endotoxemia. In macrophages, LPS tolerance (sequential LPS stimulation) demonstrated lower cell activities than the single LPS stimulation, as indicated by the reduction in supernatant cytokines, pro-inflammatory genes (iNOS and IL-1β), cell energy status (extracellular flux analysis), and enzymes of the glycolysis pathway (proteomic analysis). In conclusion, a gut barrier defect after splenectomy was vulnerable to enterocyte injury (such as DSS), which caused severe bacteremia due to defects in microbial control (asplenia) and endotoxemia-induced LPS tolerance. Hence, gut dysbiosis and gut bacterial translocation in patients with a splenectomy might be associated with systemic infection, and gut-barrier monitoring or intestinal tight-junction strengthening may be useful.
Keywords: dysbiosis; endotoxemia; gut barrier defect; splenectomy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Additional Candida albicans administration enhances the severity of dextran sulfate solution induced colitis mouse model through leaky gut-enhanced systemic inflammation and gut-dysbiosis but attenuated by Lactobacillus rhamnosus L34.Gut Microbes. 2020 May 3;11(3):465-480. doi: 10.1080/19490976.2019.1662712. Epub 2019 Sep 18. Gut Microbes. 2020. PMID: 31530137 Free PMC article.
-
Increased susceptibility to dextran sulfate-induced mucositis of iron-overload β-thalassemia mice, another endogenous cause of septicemia in thalassemia.Clin Sci (Lond). 2021 Jun 25;135(12):1467-1486. doi: 10.1042/CS20210328. Clin Sci (Lond). 2021. PMID: 34131711 Free PMC article.
-
Candida Worsens Klebsiella pneumoniae Induced-Sepsis in a Mouse Model with Low Dose Dextran Sulfate Solution through Gut Dysbiosis and Enhanced Inflammation.Int J Mol Sci. 2022 Jun 24;23(13):7050. doi: 10.3390/ijms23137050. Int J Mol Sci. 2022. PMID: 35806054 Free PMC article.
-
Uremia-Induced Gut Barrier Defect in 5/6 Nephrectomized Mice Is Worsened by Candida Administration through a Synergy of Uremic Toxin, Lipopolysaccharide, and (1➔3)-β-D-Glucan, but Is Attenuated by Lacticaseibacillus rhamnosus L34.Int J Mol Sci. 2022 Feb 24;23(5):2511. doi: 10.3390/ijms23052511. Int J Mol Sci. 2022. PMID: 35269654 Free PMC article.
-
Blood Bacteria-Free DNA in Septic Mice Enhances LPS-Induced Inflammation in Mice through Macrophage Response.Int J Mol Sci. 2022 Feb 8;23(3):1907. doi: 10.3390/ijms23031907. Int J Mol Sci. 2022. PMID: 35163830 Free PMC article.
Cited by
-
The leaky gut and the gut microbiome in sepsis - targets in research and treatment.Clin Sci (Lond). 2023 Apr 26;137(8):645-662. doi: 10.1042/CS20220777. Clin Sci (Lond). 2023. PMID: 37083032 Free PMC article.
-
Severe gut mucosal injury induces profound systemic inflammation and spleen-associated lymphoid organ response.Front Immunol. 2024 Jan 8;14:1340442. doi: 10.3389/fimmu.2023.1340442. eCollection 2023. Front Immunol. 2024. PMID: 38259439 Free PMC article.
-
Is There an Interplay between Environmental Factors, Microbiota Imbalance, and Cancer Chemotherapy-Associated Intestinal Mucositis?Pharmaceuticals (Basel). 2024 Aug 3;17(8):1020. doi: 10.3390/ph17081020. Pharmaceuticals (Basel). 2024. PMID: 39204125 Free PMC article.
-
Abnormal Blood Bacteriome, Gut Dysbiosis, and Progression to Severe Dengue Disease.Front Cell Infect Microbiol. 2022 Jun 17;12:890817. doi: 10.3389/fcimb.2022.890817. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35782108 Free PMC article.
-
Beta-Glucan from S. cerevisiae Protected AOM-Induced Colon Cancer in cGAS-Deficient Mice Partly through Dectin-1-Manipulated Macrophage Cell Energy.Int J Mol Sci. 2022 Sep 19;23(18):10951. doi: 10.3390/ijms231810951. Int J Mol Sci. 2022. PMID: 36142859 Free PMC article.
References
-
- Tantiworawit A., Dumnil S., Osataphan N., Rattanathammethee T., Hantrakool S., Chai-Adisaksopha C., Rattarittamrong E., Norasetthada L., Charoenkwan P. The Pros and Cons of Splenectomy in Transfusion Dependent Thalassemia Patient. Blood. 2018;132:4901. doi: 10.1182/blood-2018-99-111684. - DOI
MeSH terms
Substances
Grants and funding
- NRCT5-RGJ63001/Chulalongkorn university through Fundamental Fund 2565 and National Research Council of Thailand
- NRCT-N41A640076/National Research Council of Thailand with NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation
- IRN59W0004/TRF, International Network for Lupus Research, Thailand
- FF64-UoE020/the Thailand science research and innovation fund and the University of Phayao
- FF64-RIB008/the Thailand science research and innovation fund and the University of Phayao
LinkOut - more resources
Full Text Sources

