Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis
- PMID: 35163604
- PMCID: PMC8835969
- DOI: 10.3390/ijms23031683
Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis
Abstract
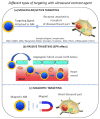
A variety of different nanomaterials (NMs) such as microbubbles (MBs), nanobubbles (NBs), nanodroplets (NDs), and silica hollow meso-structures have been tested as ultrasound contrast agents for the detection of heart diseases. The inner part of these NMs is made gaseous to yield an ultrasound contrast, which arises from the difference in acoustic impedance between the interior and exterior of such a structure. Furthermore, to specifically achieve a contrast in the diseased heart region (DHR), NMs can be designed to target this region in essentially three different ways (i.e., passively when NMs are small enough to diffuse through the holes of the vessels supplying the DHR, actively by being associated with a ligand that recognizes a receptor of the DHR, or magnetically by applying a magnetic field orientated in the direction of the DHR on a NM responding to such stimulus). The localization and resolution of ultrasound imaging can be further improved by applying ultrasounds in the DHR, by increasing the ultrasound frequency, or by using harmonic, sub-harmonic, or super-resolution imaging. Local imaging can be achieved with other non-gaseous NMs of metallic composition (i.e., essentially made of Au) by using photoacoustic imaging, thus widening the range of NMs usable for cardiac applications. These contrast agents may also have a therapeutic efficacy by carrying/activating/releasing a heart disease drug, by triggering ultrasound targeted microbubble destruction or enhanced cavitation in the DHR, for example, resulting in thrombolysis or helping to prevent heart transplant rejection.
Keywords: cancer; contrast agent; high intensity ultrasounds; nano-oncology; nanomaterials; nanomedicine; nanotechnology; sonodynamic therapy; ultrasounds.
Conflict of interest statement
Edouard Alphandéry has been working at the company Nanobacterie.
Figures



Similar articles
-
Nano-lipospheres as acoustically active ultrasound contrast agents: evolving tumor imaging and therapy technique.Nanotechnology. 2019 May 3;30(18):182001. doi: 10.1088/1361-6528/aafeb9. Epub 2019 Jan 15. Nanotechnology. 2019. PMID: 30645984 Review.
-
Ultrasound contrast agents: basic principles.Eur J Radiol. 1998 May;27 Suppl 2:S157-60. doi: 10.1016/s0720-048x(98)00057-6. Eur J Radiol. 1998. PMID: 9652516 Review.
-
Ultrasound nanotheranostics in fighting cancer: Advances and prospects.Cancer Lett. 2020 Feb 1;470:204-219. doi: 10.1016/j.canlet.2019.11.034. Epub 2019 Nov 29. Cancer Lett. 2020. PMID: 31790760 Review.
-
Microbubble Agents: New Directions.Ultrasound Med Biol. 2020 Jun;46(6):1326-1343. doi: 10.1016/j.ultrasmedbio.2020.01.027. Epub 2020 Mar 11. Ultrasound Med Biol. 2020. PMID: 32169397 Review.
-
Ultrasound-mediated diagnostic imaging and advanced treatment with multifunctional micro/nanobubbles.Cancer Lett. 2020 Apr 10;475:92-98. doi: 10.1016/j.canlet.2020.01.028. Epub 2020 Feb 4. Cancer Lett. 2020. PMID: 32032678 Review.
Cited by
-
Ultrasound-targeted microbubble destruction remodels tumour microenvironment to improve immunotherapeutic effect.Br J Cancer. 2023 Mar;128(5):715-725. doi: 10.1038/s41416-022-02076-y. Epub 2022 Dec 3. Br J Cancer. 2023. PMID: 36463323 Free PMC article. Review.
-
Noninvasive quantification of granzyme B in cardiac allograft rejection using targeted ultrasound imaging.Front Immunol. 2023 Jun 26;14:1164183. doi: 10.3389/fimmu.2023.1164183. eCollection 2023. Front Immunol. 2023. PMID: 37435082 Free PMC article.
-
Ultrasound contrast agents from microbubbles to biogenic gas vesicles.Med Rev (2021). 2022 Oct 19;3(1):31-48. doi: 10.1515/mr-2022-0020. eCollection 2023 Feb. Med Rev (2021). 2022. PMID: 37724107 Free PMC article. Review.
-
Expanding horizons in theragnostics: from oncology to multidisciplinary applications.Radiol Med. 2025 May;130(5):613-628. doi: 10.1007/s11547-025-01971-7. Epub 2025 Mar 5. Radiol Med. 2025. PMID: 40042756 Review.
-
Construction of a Tumor-Targeting Nanobubble with Multiple Scattering Interfaces and its Enhancement of Ultrasound Imaging.Int J Nanomedicine. 2024 May 22;19:4651-4665. doi: 10.2147/IJN.S462917. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38799698 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

