Gemini and Bicephalous Surfactants: A Review on Their Synthesis, Micelle Formation, and Uses
- PMID: 35163721
- PMCID: PMC8836724
- DOI: 10.3390/ijms23031798
Gemini and Bicephalous Surfactants: A Review on Their Synthesis, Micelle Formation, and Uses
Abstract
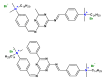
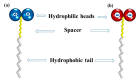
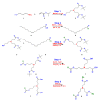
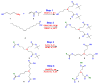
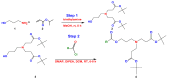
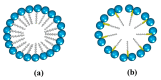
The use of surfactants in polymerization reactions is particularly important, mainly in emulsion polymerizations. Further, micelles from biocompatible surfactants find use in pharmaceutical dosage forms. This paper reviews recent developments in the synthesis of novel gemini and bicephalous surfactants, micelle formation, and their applications in polymer and nanoparticle synthesis, oil recovery, catalysis, corrosion, protein binding, and biomedical area, particularly in drug delivery.
Keywords: amphiphiles; bicephalous surfactants; drug nanocarriers; gemini surfactants; micelles; polymer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

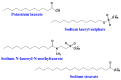
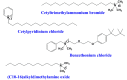
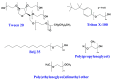
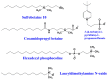









Similar articles
-
On the Spacer Group Effect on Critical Micelle Concentration of Cationic Gemini Surfactants Using Molecular Connectivity Indices.Comb Chem High Throughput Screen. 2016;19(6):481-8. doi: 10.2174/1386207319666160504095717. Comb Chem High Throughput Screen. 2016. PMID: 27141990
-
Dissymmetric gemini surfactants generated by disulfide exchange in mixed micelles.J Oleo Sci. 2010;59(2):95-100. doi: 10.5650/jos.59.95. J Oleo Sci. 2010. PMID: 20103982
-
Synthesis and aggregation properties of dissymmetric phytanyl-gemini surfactants for use as improved DNA transfection vectors.Phys Chem Chem Phys. 2011 Jan 14;13(2):637-42. doi: 10.1039/c0cp00146e. Epub 2010 Oct 28. Phys Chem Chem Phys. 2011. PMID: 20981388
-
Ionic Liquid-Based Surfactants: Recent Advances in Their Syntheses, Solution Properties, and Applications.Polymers (Basel). 2021 Mar 30;13(7):1100. doi: 10.3390/polym13071100. Polymers (Basel). 2021. PMID: 33808369 Free PMC article. Review.
-
Recent advances in gemini surfactants: oleic Acid-based gemini surfactants and polymerizable gemini surfactants.J Oleo Sci. 2011;60(4):159-63. doi: 10.5650/jos.60.159. J Oleo Sci. 2011. PMID: 21427511 Review.
Cited by
-
Ferroptosis in Haematological Malignancies and Associated Therapeutic Nanotechnologies.Int J Mol Sci. 2023 Apr 21;24(8):7661. doi: 10.3390/ijms24087661. Int J Mol Sci. 2023. PMID: 37108836 Free PMC article. Review.
-
Enhancing cancer therapy: advanced nanovehicle delivery systems for oridonin.Front Pharmacol. 2024 Dec 3;15:1476739. doi: 10.3389/fphar.2024.1476739. eCollection 2024. Front Pharmacol. 2024. PMID: 39691396 Free PMC article. Review.
-
Synthesis of New Cationic Dicephalic Surfactants and Their Nonequivalent Adsorption at the Air/Solution Interface.Langmuir. 2025 Apr 1;41(12):8125-8137. doi: 10.1021/acs.langmuir.4c04803. Epub 2025 Mar 21. Langmuir. 2025. PMID: 40116631 Free PMC article.
-
Novel Gemini Surfactant for Binding Eu(III)-Polyoxometalate into Hydrogels and Polymer Latexes.Gels. 2022 Nov 30;8(12):786. doi: 10.3390/gels8120786. Gels. 2022. PMID: 36547310 Free PMC article.
-
The influence of a dicationic surfactant on the aggregation process of the IVAGVN peptide derived from the human cystatin C sequence (56-61).RSC Adv. 2025 Jan 31;15(5):3237-3249. doi: 10.1039/d4ra08377f. eCollection 2025 Jan 29. RSC Adv. 2025. PMID: 39896427 Free PMC article.
References
-
- MacNaught A.D., Wilkinson A.R. Compendium of Chemical Terminology: IUPAC Recommendations. 2nd ed. Wiley; Hoboken, NJ, USA: 1997.
-
- Slomkowski S., Alemán J.V., Gilbert R.G., Hess M., Horie K., Jones R.G., Kubisa P., Meisel I., Mormann W., Penczek S., et al. Terminology of polymers and polymerization processes in dispersed systems (IUPAC Recommendations 2011) Pure Appl. Chem. 2011;83:2229–2259. doi: 10.1351/PAC-REC-10-06-03. - DOI
-
- Lombardo D., Kiselev M.A., Magazù S., Calandra P. Amphiphiles self-assembly: Basic concepts and future perspectives of supramolecular approaches. Adv. Condens. Matter Phys. 2015;151683:1–22. doi: 10.1155/2015/151683. - DOI
-
- Myers D. Surfactant Science and Technology. 3rd ed. John Wiley & Sons; Hoboken, NJ, USA: 2005.
-
- Schramm L.L. Surfactants: Fundamentals and Applications in the Petroleum Industry. Cambridge University Press; New York, NY, USA: 2000.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

