Recent Advances in the Chemistry and Therapeutic Evaluation of Naturally Occurring and Synthetic Withanolides
- PMID: 35164150
- PMCID: PMC8840339
- DOI: 10.3390/molecules27030886
Recent Advances in the Chemistry and Therapeutic Evaluation of Naturally Occurring and Synthetic Withanolides
Abstract
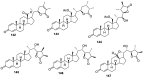
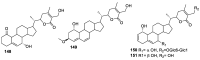
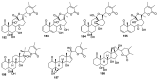
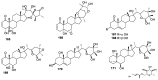
Natural products are a major source of biologically active compounds that make promising lead molecules for developing efficacious drug-like molecules. Natural withanolides are found in many flora and fauna, including plants, algae, and corals, that traditionally have shown multiple health benefits and are known for their anti-cancer, anti-inflammatory, anti-bacterial, anti-leishmaniasis, and many other medicinal properties. Structures of these withanolides possess a few reactive sites that can be exploited to design and synthesize more potent and safe analogs. In this review, we discuss the literature evidence related to the medicinal implications, particularly anticancer properties of natural withanolides and their synthetic analogs, and provide perspectives on the translational potential of these promising compounds.
Keywords: anti-bacterial; anti-inflammatory; anti-leishmaniasis; anticancer; withaferin A; withanolides.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Figures


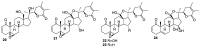
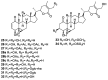

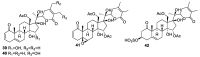
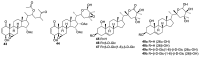
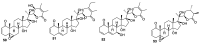
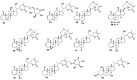
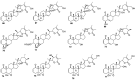


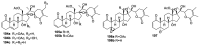
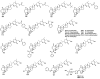
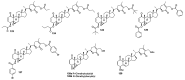
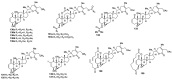
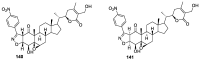


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

