Signal Peptidase-Mediated Cleavage of the Anti-σ Factor RsiP at Site 1 Controls σP Activation and β-Lactam Resistance in Bacillus thuringiensis
- PMID: 35164554
- PMCID: PMC8844934
- DOI: 10.1128/mbio.03707-21
Signal Peptidase-Mediated Cleavage of the Anti-σ Factor RsiP at Site 1 Controls σP Activation and β-Lactam Resistance in Bacillus thuringiensis
Abstract
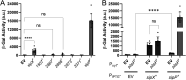
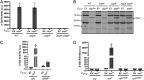
In Bacillus thuringiensis, β-lactam antibiotic resistance is controlled by the extracytoplasmic function (ECF) σ factor σP. σP activity is inhibited by the anti-σ factor RsiP. In the presence of β-lactam antibiotics, RsiP is degraded and σP is activated. Previous work found that RsiP degradation requires cleavage of RsiP at site 1 by an unknown protease, followed by cleavage at site 2 by the site 2 protease RasP. The penicillin-binding protein PbpP acts as a sensor for β-lactams. PbpP initiates σP activation and is required for site 1 cleavage of RsiP but is not the site 1 protease. Here, we describe the identification of a signal peptidase, SipP, which cleaves RsiP at a site 1 signal peptidase cleavage site and is required for σP activation. Finally, many B. anthracis strains are sensitive to β-lactams yet encode the σP-RsiP signal transduction system. We identified a naturally occurring mutation in the signal peptidase cleavage site of B. anthracis RsiP that renders it resistant to SipP cleavage. We find that B. anthracis RsiP is not degraded in the presence of β-lactams. Altering the B. anthracis RsiP site 1 cleavage site by a single residue to resemble B. thuringiensis RsiP results in β-lactam-dependent degradation of RsiP. We show that mutation of the B. thuringiensis RsiP cleavage site to resemble the sequence of B. anthracis RsiP blocks degradation by SipP. The change in the cleavage site likely explains many reasons why B. anthracis strains are sensitive to β-lactams. IMPORTANCE β-Lactam antibiotics are important for the treatment of many bacterial infections. However, resistance mechanisms have become increasingly more prevalent. Understanding how β-lactam resistance is conferred and how bacteria control expression of β-lactam resistance is important for informing the future treatment of bacterial infections. σP is an alternative σ factor that controls the transcription of genes that confer β-lactam resistance in Bacillus thuringiensis, Bacillus cereus, and Bacillus anthracis. Here, we identify a signal peptidase as the protease required for initiating activation of σP by the degradation of the anti-σ factor RsiP. The discovery that the signal peptidase SipP is required for σP activation highlights an increasing role for signal peptidases in signal transduction, as well as in antibiotic resistance.
Keywords: cell envelope; gene expression; sigma factors; signal transduction; stress response; σ factors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
