CPR63 promotes pyrethroid resistance by increasing cuticle thickness in Culex pipiens pallens
- PMID: 35164827
- PMCID: PMC8842966
- DOI: 10.1186/s13071-022-05175-0
CPR63 promotes pyrethroid resistance by increasing cuticle thickness in Culex pipiens pallens
Abstract
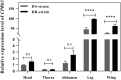
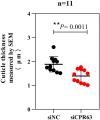
The cuticle protein (CP) encoded by CPR63 plays a role in deltamethrin resistance in Culex pipiens pallens. Herein, we investigated the distribution of CPR63 transcripts in this organism and observed high expression levels in legs and wings. Furthermore, expression of CPR63 in the legs of deltamethrin-resistant (DR) strains was 2.17-fold higher than in deltamethrin-susceptible (DS) strains. Cuticle analysis of small interfering RNA (siRNA) groups by scanning electron microscopy (SEM) revealed a significantly thinner cuticle of the tarsi in the siCPR63 group than in the siNC (negative control siRNA) group. Transmission electron microscopy (TEM) revealed that the exocuticle and endocuticle thickness of the tarsi were significantly thinner, which contributes the thinner procuticle of tarsi in the siCPR63 group than in the siNC group. Our results suggested that CPR63 might contribute to the resistance phenotype by thickening the cuticle and thereby possibly increasing the tolerance of mosquitoes to deltamethrin.
Keywords: CPRs; Culex pipiens pallens; Cuticle thickening; Cuticular resistance; Deltamethrin; Mosquito.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
Transcription factor FTZ-F1 regulates mosquito cuticular protein CPLCG5 conferring resistance to pyrethroids in Culex pipiens pallens.Parasit Vectors. 2020 Oct 14;13(1):514. doi: 10.1186/s13071-020-04383-w. Parasit Vectors. 2020. PMID: 33054862 Free PMC article.
-
Culex pipiens pallens cuticular protein CPLCG5 participates in pyrethroid resistance by forming a rigid matrix.Parasit Vectors. 2018 Jan 4;11(1):6. doi: 10.1186/s13071-017-2567-9. Parasit Vectors. 2018. PMID: 29301564 Free PMC article.
-
The cuticle proteins: a putative role for deltamethrin resistance in Culex pipiens pallens.Parasitol Res. 2015 Dec;114(12):4421-9. doi: 10.1007/s00436-015-4683-9. Epub 2015 Sep 4. Parasitol Res. 2015. PMID: 26337265
-
Cuticle genes CpCPR63 and CpCPR47 may confer resistance to deltamethrin in Culex pipiens pallens.Parasitol Res. 2017 Aug;116(8):2175-2179. doi: 10.1007/s00436-017-5521-z. Epub 2017 Jun 12. Parasitol Res. 2017. PMID: 28608057
-
Pyrethroid resistance in Culex pipiens mosquitoes.Pestic Biochem Physiol. 2015 May;120:68-76. doi: 10.1016/j.pestbp.2014.12.018. Epub 2014 Dec 19. Pestic Biochem Physiol. 2015. PMID: 25987223 Review.
Cited by
-
Transcriptomic analysis of Anopheles gambiae from Benin reveals overexpression of salivary and cuticular proteins associated with cross-resistance to pyrethroids and organophosphates.BMC Genomics. 2024 Apr 6;25(1):348. doi: 10.1186/s12864-024-10261-x. BMC Genomics. 2024. PMID: 38582836 Free PMC article.
-
Ultrastructure and Transcriptome Analysis of the Larval Integument in Solitary and Gregarious Phases of Mythimna separata.Insects. 2025 Feb 10;16(2):190. doi: 10.3390/insects16020190. Insects. 2025. PMID: 40003822 Free PMC article.
-
Transcriptome Analysis Unveils Molecular Mechanisms of Acaricide Resistance in Two-Spotted Spider Mite Populations on Hops.Int J Mol Sci. 2024 Dec 11;25(24):13298. doi: 10.3390/ijms252413298. Int J Mol Sci. 2024. PMID: 39769060 Free PMC article.
-
A Leg Cuticle Protein Enhances the Resistance of Anopheles sinensis Mosquitoes to Deltamethrin.Int J Mol Sci. 2025 Feb 28;26(5):2182. doi: 10.3390/ijms26052182. Int J Mol Sci. 2025. PMID: 40076801 Free PMC article.
-
Key gene modules and hub genes associated with pyrethroid and organophosphate resistance in Anopheles mosquitoes: a systems biology approach.BMC Genomics. 2024 Jul 3;25(1):665. doi: 10.1186/s12864-024-10572-z. BMC Genomics. 2024. PMID: 38961324 Free PMC article.
References
-
- WHO P : Word malaria report 2020. World Health Organization 2020.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

