Potassium Aluminyl Promoted Carbonylation of Ethene
- PMID: 35166007
- PMCID: PMC9307019
- DOI: 10.1002/anie.202117396
Potassium Aluminyl Promoted Carbonylation of Ethene
Abstract
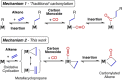
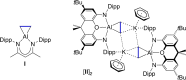
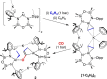
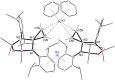
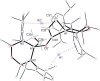
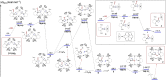
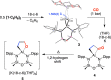
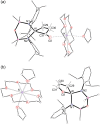
The potassium aluminyl [K{Al(NONDipp )}]2 ([NONDipp ]2- =[O{SiMe2 NDipp}2 ]2- , Dipp=2,6-iPr2 C6 H3 ) activates ethene towards carbonylation with CO under mild conditions. An isolated bis-aluminacyclopropane compound reacted with CO via carbonylation of an Al-C bond, followed by an intramolecular hydrogen shift to form K2 [Al(NONDipp )(μ-CH2 CH=CO-1κ2 C1,3 -2κO)Al(NONDipp )Et]. Restricting the chemistry to a mono-aluminium system allowed isolation of [Al(NONDipp )(CH2 CH2 CO-κ2 C1,3 )]- , which undergoes thermal isomerisation to form the [Al(NONDipp )(CH2 CH=CHO-κ2 C,O)]- anion. DFT calculations highlight the stabilising influence of incorporated benzene at multiple steps in the reaction pathways.
Keywords: Aluminyl Anions; Bimetallic Complexes; Carbonylation; Hydrogen-Shift; Metallacycles.
© 2022 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Carbon-Carbon Bond Forming Reactions Promoted by Aluminyl and Alumoxane Anions: Introducing the Ethenetetraolate Ligand.Angew Chem Int Ed Engl. 2020 Jul 27;59(31):12806-12810. doi: 10.1002/anie.202005301. Epub 2020 Jun 2. Angew Chem Int Ed Engl. 2020. PMID: 32378311
-
Dihydrogen Activation by Lithium- and Sodium-Aluminyls.Angew Chem Int Ed Engl. 2021 Oct 4;60(41):22289-22292. doi: 10.1002/anie.202108934. Epub 2021 Aug 31. Angew Chem Int Ed Engl. 2021. PMID: 34402149
-
Reductive Coupling of a Diazoalkane Derivative Promoted by a Potassium Aluminyl and Elimination of Dinitrogen to Generate a Reactive Aluminium Ketimide.Chemistry. 2023 Dec 19;29(71):e202302903. doi: 10.1002/chem.202302903. Epub 2023 Nov 7. Chemistry. 2023. PMID: 37786384 Free PMC article.
-
Calcium-Catalyzed Arene C-H Bond Activation by Low-Valent AlI.Angew Chem Int Ed Engl. 2019 Oct 21;58(43):15496-15503. doi: 10.1002/anie.201908978. Epub 2019 Sep 13. Angew Chem Int Ed Engl. 2019. PMID: 31465144 Free PMC article. Review.
-
The emerging chemistry of the aluminyl anion.Chem Commun (Camb). 2023 Jan 12;59(5):503-519. doi: 10.1039/d2cc05963k. Chem Commun (Camb). 2023. PMID: 36541674 Review.
Cited by
-
Alumanyl Reduction, Reductive Coupling and C-H Isomerization of Organic Nitriles.Organometallics. 2024 Aug 16;43(17):1938-1945. doi: 10.1021/acs.organomet.4c00289. eCollection 2024 Sep 9. Organometallics. 2024. PMID: 39268183 Free PMC article.
-
Terminal Alkyne Activation by an Al(I)-Centered Anion: Impact on the Mechanism of Alkali Metal Identity.Organometallics. 2024 Dec 9;44(1):236-243. doi: 10.1021/acs.organomet.4c00435. eCollection 2025 Jan 13. Organometallics. 2024. PMID: 39822185 Free PMC article.
-
Allosteric Differentiation of Al(I) Reactivity.Chemistry. 2025 May 27;31(30):e202501352. doi: 10.1002/chem.202501352. Epub 2025 Apr 25. Chemistry. 2025. PMID: 40211470 Free PMC article.
-
Assembly of Functionalized Organic Fragments via Reductive Activation and (Cross)-Coupling of C2H4, CO, CO2 and/or H2 Using a Magnesium-Dinitrogen Complex.Angew Chem Int Ed Engl. 2025 May;64(19):e202500264. doi: 10.1002/anie.202500264. Epub 2025 Mar 12. Angew Chem Int Ed Engl. 2025. PMID: 40016175 Free PMC article.
-
Controlled reductive C-C coupling of isocyanides promoted by an aluminyl anion.Chem Sci. 2023 May 12;14(23):6278-6288. doi: 10.1039/d3sc01387a. eCollection 2023 Jun 14. Chem Sci. 2023. PMID: 37325153 Free PMC article.
References
-
- Peng J.-B., Geng H.-Q., Wu X.-F., Chem 2019, 5, 526–552.
-
- None
-
- Franke R., Selent D., Börner A., Chem. Rev. 2012, 112, 5675–5732; - PubMed
-
- Wiese K.-D., Obst D., in: Topics in Organometallic Chemistry (Ed.: Beller M.), Springer, Berlin, Heidelberg, 2006, pp. 1–33.
-
- El Ali B., Alper H., in: Transition Metals for Organic Synthesis, Vol. 1 (Eds.: Beller M., Bolm C.), Wiley-VCH, Weinheim, 2004.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

