Modular head-mounted cortical imaging device for chronic monitoring of intrinsic signals in mice
- PMID: 35166087
- PMCID: PMC8843356
- DOI: 10.1117/1.JBO.27.2.026501
Modular head-mounted cortical imaging device for chronic monitoring of intrinsic signals in mice
Abstract
Significance: Intrinsic optical signals (IOS) generated in the cortical tissue as a result of various interacting metabolic processes are used extensively to elucidate the underlying mechanisms that govern neurovascular coupling. However, current IOS measurements still often rely on bulky, tabletop imaging systems, and there remains a dearth of studies in freely moving subjects. Lightweight, miniature head-mounted imaging devices provide unique opportunities for investigating cortical dynamics in small animals under a variety of naturalistic behavioral settings.
Aim: The aim of this work was to monitor IOS in the somatosensory cortex of wild-type mice by developing a lightweight, biocompatible imaging device that readily lends itself to animal experiments in freely moving conditions.
Approach: Herein we describe a method for realizing long-term IOS imaging in mice using a 0.54-g, compact, CMOS-based, head-mounted imager. The two-part module, consisting of a tethered sensor plate and a base plate, allows facile assembly prior to imaging sessions and disassembly when the sensor is not in use. LEDs integrated into the device were chosen to illuminate the cortical mantle at two different wavelengths in the visible regime (λcenter: 535 and 625 nm) for monitoring volume- and oxygenation state-dependent changes in the IOS, respectively. To test whether the system can detect robust cortical responses, we recorded sensory-evoked IOS from mechanical stimulation of the hindlimbs (HL) of anesthetized mice in both acute and long-term implantation conditions.
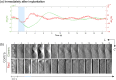
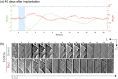
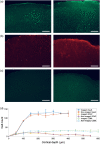
Results: Cortical IOS recordings in the primary somatosensory cortex hindlimb receptive field (S1HL) of anesthetized mice under green and red LED illumination revealed robust, multiphasic profiles that were time-locked to the mechanical stimulation of the contralateral plantar hindpaw. Similar intrinsic signal profiles observed in S1HL at 40 days postimplantation demonstrated the viability of the approach for long-term imaging. Immunohistochemical analysis showed that the brain tissue did not exhibit appreciable immune response due to the device implantation and operation. A proof-of-principle imaging session in a freely behaving mouse showed minimal locomotor impediment for the animal and also enabled estimation of blood flow speed.
Conclusions: We demonstrate the utility of a miniature cortical imaging device for monitoring IOS and related hemodynamic processes in both anesthetized and freely moving mice, cueing potential for applications to some neuroscientific studies of sensation and naturalistic behavior.
Keywords: CMOS; chronic imaging; cortical imaging; head-mounted devices; intrinsic optical signal.
Figures



References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

