Imaging-guided bioreactor for de-epithelialization and long-term cultivation of ex vivo rat trachea
- PMID: 35166739
- PMCID: PMC8942046
- DOI: 10.1039/d1lc01105g
Imaging-guided bioreactor for de-epithelialization and long-term cultivation of ex vivo rat trachea
Abstract
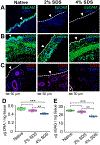
Recent synergistic advances in organ-on-chip and tissue engineering technologies offer opportunities to create in vitro-grown tissue or organ constructs that can faithfully recapitulate their in vivo counterparts. Such in vitro tissue or organ constructs can be utilized in multiple applications, including rapid drug screening, high-fidelity disease modeling, and precision medicine. Here, we report an imaging-guided bioreactor that allows in situ monitoring of the lumen of ex vivo airway tissues during controlled in vitro tissue manipulation and cultivation of isolated rat trachea. Using this platform, we demonstrated partial removal of the rat tracheal epithelium (i.e., de-epithelialization) without disrupting the underlying subepithelial cells and extracellular matrix. Through different tissue evaluation assays, such as immunofluorescent staining, DNA/protein quantification, and electron beam microscopy, we showed that the epithelium of the tracheal lumen can be effectively removed with negligible disruption in the underlying tissue layers, such as cartilage and blood vessel. Notably, using a custom-built micro-optical imaging device integrated with the bioreactor, the trachea lumen was visualized at the cellular level, and removal of the endogenous epithelium and distribution of locally delivered exogenous cells were demonstrated in situ. Moreover, the de-epithelialized trachea supported on the bioreactor allowed attachment and growth of exogenous cells seeded topically on its denuded tissue surface. Collectively, the results suggest that our imaging-enabled rat trachea bioreactor and localized cell replacement method can facilitate creation of bioengineered in vitro airway tissue that can be used in different biomedical applications.
Conflict of interest statement
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
Imaging-Guided Bioreactor for Generating Bioengineered Airway Tissue.J Vis Exp. 2022 Apr 6;(182):10.3791/63544. doi: 10.3791/63544. J Vis Exp. 2022. PMID: 35467661 Free PMC article.
-
Homogeneous Distribution of Exogenous Cells onto De-epithelialized Rat Trachea via Instillation of Cell-Loaded Hydrogel.ACS Biomater Sci Eng. 2022 Jan 10;8(1):82-88. doi: 10.1021/acsbiomaterials.1c01031. Epub 2021 Dec 7. ACS Biomater Sci Eng. 2022. PMID: 34874712 Free PMC article.
-
Bioreactor-Based De-epithelialization of Long-Segment Tracheal Grafts.Methods Mol Biol. 2022;2436:167-182. doi: 10.1007/7651_2021_431. Methods Mol Biol. 2022. PMID: 34505268
-
Tissue engineering technologies: just a quick note about transplantation of bioengineered donor trachea and augmentation cystoplasty by de novo engineered bladder tissue.G Chir. 2009 Nov-Dec;30(11-12):514-9. G Chir. 2009. PMID: 20109384 Review.
-
Tissue-engineered trachea: A review.Int J Pediatr Otorhinolaryngol. 2016 Dec;91:55-63. doi: 10.1016/j.ijporl.2016.10.012. Epub 2016 Oct 14. Int J Pediatr Otorhinolaryngol. 2016. PMID: 27863642 Review.
Cited by
-
A Cell State Monitoring System with Integrated In Situ Imaging and pH Detection.Sensors (Basel). 2023 Nov 22;23(23):9340. doi: 10.3390/s23239340. Sensors (Basel). 2023. PMID: 38067713 Free PMC article.
-
In situ vascularization and epithelialization of segmental bioengineered trachea based on marrow-derived stem/progenitor cells.Mater Today Bio. 2025 Jun 14;33:101990. doi: 10.1016/j.mtbio.2025.101990. eCollection 2025 Aug. Mater Today Bio. 2025. PMID: 40605986 Free PMC article.
-
Bioengineered tracheal graft with enhanced vascularization and mechanical stability for functional airway reconstruction.Regen Ther. 2025 Apr 9;29:364-380. doi: 10.1016/j.reth.2025.03.016. eCollection 2025 Jun. Regen Ther. 2025. PMID: 40248768 Free PMC article. Review.
-
Opto-electromechanical quantification of epithelial barrier function in injured and healthy airway tissues.APL Bioeng. 2023 Jan 11;7(1):016104. doi: 10.1063/5.0123127. eCollection 2023 Mar. APL Bioeng. 2023. PMID: 36644417 Free PMC article.
-
Development and characterization of ferret ex vivo tracheal injury and cell engraftment model.Front Med (Lausanne). 2023 Apr 11;10:1144754. doi: 10.3389/fmed.2023.1144754. eCollection 2023. Front Med (Lausanne). 2023. PMID: 37113613 Free PMC article.
References
-
- Wanner A, Salathé M and O'Riordan TG, Am. J. Respir. Crit. Care Med, 1996, 154, 1868–1902. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

