Assessing the effects of stress on feeding behaviors in laboratory mice
- PMID: 35167441
- PMCID: PMC8846584
- DOI: 10.7554/eLife.70271
Assessing the effects of stress on feeding behaviors in laboratory mice
Abstract
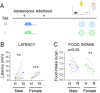
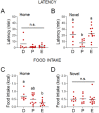
Stress often affects eating behaviors, increasing caloric intake in some individuals and decreasing it in others. The determinants of feeding responses to stress are unknown, in part because this issue is rarely studied in rodents. We focused our efforts on the novelty-suppressed feeding (NSF) assay, which uses latency to eat as readout of anxiety-like behavior, but rarely assesses feeding per se. We explored how key variables in experimental paradigms - estrous and diurnal cyclicity, age and duration of social isolation, prandial state, diet palatability, and elevated body weight - influence stress-induced anxiety-like behavior and food intake in male and female C57BL/6J mice. Latency to eat in the novel environment is increased in both sexes across most of the conditions tested, while effects on caloric intake are variable. In the common NSF assay (i.e., lean mice in the light cycle), sex-specific effects of the length of social isolation, and not estrous cyclicity, are the main source of variability. Under conditions that are more physiologically relevant for humans (i.e., overweight mice in the active phase), the novel stress now elicits robust hyperphagia in both sexes . This novel model of stress eating can be used to identify underlying neuroendocrine and neuronal substrates. Moreover, these studies can serve as a framework to integrate cross-disciplinary studies of anxiety and feeding related behaviors in rodents.
Keywords: circadian rhythm; emotional eating; mouse; neuroscience; novelty suppressed feeding; sex differences; social isolation; stress.
Plain language summary
In times of heightened anxiety – say, during a global pandemic – many of us will reach for donuts or a particularly appetizing pizza for comfort. Others, however, will tend to shun food. What underlies these differences, and, in fact, the neural and hormonal pathways at play during stress eating (when people eat without being hungry due to emotional reasons), remain unclear. This is partly because scientists lack good animal models in which to study these behaviors. In particular, female rodents are usually excluded from studies under the assumption that their hormonal cycles will disrupt the results. Yet, women are overrepresented in studies on feeding habits. Modeling human behaviors using rodents is harder than it may appear. These animals are most active at night – yet most experiments are performed during the day. The same stressors also have different outcomes in males and females. François et al. therefore explored better ways to induce anxiety and evaluate feeding behavior in mice, hoping to reliably elicit stress eating. The starting point was a common type of experiments known as novelty-suppressed feeding. First, mice are kept alone in a cage for up to two weeks on a normal diet so that they are used to experimental conditions. Then they are deprived of food overnight, before being given free access to food in the morning in a new environment. This stressful experience normally causes mice to take longer to eat than in their home cage. In rodents, the delay is thought to reflect stress as it is reliably reversed by anti-anxiety compounds approved for human use. In the novelty-suppressed feeding assay, both male and female animals exhibit signs of anxiety, but how much females eat is variable. François et al. showed that this variability is not due to hormonal changes, but instead to how long female mice had been kept alone. Crucially, the test could be adapted so that mice would consistently exhibit behavior similar to human stress eating, whereby they eat more during the test without having fasted the night before. The changes included running the experiment at night, when the animals are normally most active, and using overweight mice (which captures the fact that, in humans, being overweight is associated with being prone to stress eating). Stress eating is an important clinical issue, hindering weigh loss in people with obesity. The new model developed by François et al. could be adopted by other laboratories, enabling better research into this behavior.
© 2022, Francois et al.
Conflict of interest statement
MF, IC, NS, CL, LZ No competing interests declared
Figures











References
-
- Bartolomucci A, Cabassi A, Govoni P, Ceresini G, Cero C, Berra D, Dadomo H, Franceschini P, Dell’Omo G, Parmigiani S, Palanza P, Baune B. Metabolic Consequences and Vulnerability to Diet-Induced Obesity in Male Mice under Chronic Social Stress. PLOS ONE. 2009;4:e4331. doi: 10.1371/journal.pone.0004331. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

