A systematic review and meta-analyses of interleukin-1 receptor associated kinase 3 (IRAK3) action on inflammation in in vivo models for the study of sepsis
- PMID: 35167625
- PMCID: PMC8846508
- DOI: 10.1371/journal.pone.0263968
A systematic review and meta-analyses of interleukin-1 receptor associated kinase 3 (IRAK3) action on inflammation in in vivo models for the study of sepsis
Abstract
Background: Interleukin-1 receptor associated kinase 3 (IRAK3) is a critical modulator of inflammation and is associated with endotoxin tolerance and sepsis. Although IRAK3 is known as a negative regulator of inflammation, several studies have reported opposing functions, and the temporal actions of IRAK3 on inflammation remain unclear. A systematic review and meta-analyses were performed to investigate IRAK3 expression and its effects on inflammatory markers (TNF-α and IL-6) after one- or two-challenge interventions, which mimic the hyperinflammatory and immunosuppression phases of sepsis, respectively, using human or animal in vivo models.
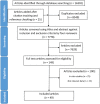
Methods: This systematic review and meta-analyses has been registered in the Open Science Framework (OSF) (Registration DOI: 10.17605/OSF.IO/V39UR). A systematic search was performed to identify in vivo studies reporting outcome measures of expression of IRAK3 and inflammatory markers. Meta-analyses were performed where sufficient data was available.
Results: The search identified 7778 studies for screening. After screening titles, abstracts and full texts, a total of 49 studies were included in the systematic review. The review identified significant increase of IRAK3 mRNA and protein expression at different times in humans compared to rodents following one-challenge, whereas the increases of IL-6 and TNF-α protein expression in humans were similar to rodent in vivo models. Meta-analyses confirmed the inhibitory effect of IRAK3 on TNF-α mRNA and protein expression after two challenges.
Conclusions: A negative correlation between IRAK3 and TNF-α expression in rodents following two challenges demonstrates the association of IRAK3 in the immunosuppression phase of sepsis. Species differences in underlying biology affect the translatability of immune responses of animal models to human, as shown by the dissimilarity in patterns of IRAK3 mRNA and protein expression between humans and rodents following one challenge that are further influenced by variations in experimental procedures.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures







Similar articles
-
Analysis of interleukin-1 receptor associated kinase-3 (IRAK3) function in modulating expression of inflammatory markers in cell culture models: A systematic review and meta-analysis.PLoS One. 2020 Dec 31;15(12):e0244570. doi: 10.1371/journal.pone.0244570. eCollection 2020. PLoS One. 2020. PMID: 33382782 Free PMC article.
-
IRAK3 Knockout and Wildtype THP-1 Monocytes as Models for Endotoxin Detection Assays and Fusobacterium nucleatum Bacteriophage FNU1 Cytokine Induction.Int J Mol Sci. 2023 Oct 12;24(20):15108. doi: 10.3390/ijms242015108. Int J Mol Sci. 2023. PMID: 37894788 Free PMC article.
-
Downregulation of IRAK3 by miR-33b-3p relieves chondrocyte inflammation and apoptosis in an in vitro osteoarthritis model.Biosci Biotechnol Biochem. 2021 Feb 24;85(3):545-552. doi: 10.1093/bbb/zbaa105. Biosci Biotechnol Biochem. 2021. PMID: 33590831
-
Interleukin-1 receptor-associated kinase 3 gene associates with susceptibility to acute lung injury.Am J Respir Cell Mol Biol. 2011 Oct;45(4):740-5. doi: 10.1165/rcmb.2010-0292OC. Epub 2011 Feb 4. Am J Respir Cell Mol Biol. 2011. PMID: 21297081 Free PMC article.
-
Can coenzyme Q10 supplementation effectively reduce human tumor necrosis factor-α and interleukin-6 levels in chronic inflammatory diseases? A systematic review and meta-analysis of randomized controlled trials.Pharmacol Res. 2019 Oct;148:104290. doi: 10.1016/j.phrs.2019.104290. Epub 2019 Jun 8. Pharmacol Res. 2019. PMID: 31185284
Cited by
-
3D Hydrogel Culture System Recapitulates Key Tuberculosis Phenotypes and Demonstrates Pyrazinamide Efficacy.Adv Healthc Mater. 2025 Feb;14(5):e2304299. doi: 10.1002/adhm.202304299. Epub 2024 May 1. Adv Healthc Mater. 2025. PMID: 38655817
-
Elucidating common pathogenic transcriptional networks in infective endocarditis and sepsis: integrated insights from biomarker discovery and single-cell RNA sequencing.Front Immunol. 2024 Jan 25;14:1298041. doi: 10.3389/fimmu.2023.1298041. eCollection 2023. Front Immunol. 2024. PMID: 38332910 Free PMC article.
-
The IRAK-M death domain: a tale of three surfaces.Front Mol Biosci. 2024 Jan 10;10:1265455. doi: 10.3389/fmolb.2023.1265455. eCollection 2023. Front Mol Biosci. 2024. PMID: 38268724 Free PMC article.
-
Deciphering the molecular classification of pediatric sepsis: integrating WGCNA and machine learning-based classification with immune signatures for the development of an advanced diagnostic model.Front Genet. 2024 Jan 29;15:1294381. doi: 10.3389/fgene.2024.1294381. eCollection 2024. Front Genet. 2024. PMID: 38348451 Free PMC article.
-
Targeting GLI1 and BAX by nanonoscapine could impede prostate adenocarcinoma progression.Sci Rep. 2024 Aug 16;14(1):18977. doi: 10.1038/s41598-024-65968-4. Sci Rep. 2024. PMID: 39152150 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

