Insights into POT1 structural dynamics revealed by cryo-EM
- PMID: 35176105
- PMCID: PMC8853558
- DOI: 10.1371/journal.pone.0264073
Insights into POT1 structural dynamics revealed by cryo-EM
Abstract
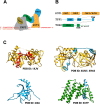
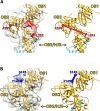
Telomeres are protein-DNA complexes that protect the ends of linear eukaryotic chromosomes. Mammalian telomeric DNA consists of 5'-(TTAGGG)n-3' double-stranded repeats, followed by up to several hundred bases of a 3' single-stranded G-rich overhang. The G-rich overhang is bound by the shelterin component POT1 which interacts with TPP1, the component involved in telomerase recruitment. A previously published crystal structure of the POT1 N-terminal half bound to the high affinity telomeric ligand 5'-TTAGGGTTAG-3' showed that the first six nucleotides, TTAGGG, are bound by the OB1 fold, while the adjacent OB2 binds the last four, TTAG. Here, we report two cryo-EM structures of full-length POT1 bound by the POT1-binding domain of TPP1. The structures differ in the relative orientation of the POT1 OB1 and OB2, suggesting that these two DNA-binding OB folds take up alternative conformations. Supporting DNA binding studies using telomeric ligands in which the OB1 and OB2 binding sites were spaced apart, show that POT1 binds with similar affinities to spaced or contiguous binding sites, suggesting plasticity in DNA binding and a role for the alternative conformations observed. A likely explanation is that the structural flexibility of POT1 enhances binding to the tandemly arranged telomeric repeats and hence increases telomere protection.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
The OB-fold domain 1 of human POT1 recognizes both telomeric and non-telomeric DNA motifs.Biochimie. 2015 Aug;115:17-27. doi: 10.1016/j.biochi.2015.04.015. Epub 2015 Apr 28. Biochimie. 2015. PMID: 25934589 Free PMC article.
-
TPP1 is a homologue of ciliate TEBP-beta and interacts with POT1 to recruit telomerase.Nature. 2007 Feb 1;445(7127):559-62. doi: 10.1038/nature05469. Epub 2007 Jan 21. Nature. 2007. PMID: 17237767
-
Coordinated interactions of multiple POT1-TPP1 proteins with telomere DNA.J Biol Chem. 2013 Jun 7;288(23):16361-16370. doi: 10.1074/jbc.M113.471896. Epub 2013 Apr 24. J Biol Chem. 2013. PMID: 23616058 Free PMC article.
-
Structural biology of telomeres and telomerase.Cell Mol Life Sci. 2020 Jan;77(1):61-79. doi: 10.1007/s00018-019-03369-x. Epub 2019 Nov 14. Cell Mol Life Sci. 2020. PMID: 31728577 Free PMC article. Review.
-
Shelterin: the protein complex that shapes and safeguards human telomeres.Genes Dev. 2005 Sep 15;19(18):2100-10. doi: 10.1101/gad.1346005. Genes Dev. 2005. PMID: 16166375 Review.
Cited by
-
CST-Polα/Primase: the second telomere maintenance machine.Genes Dev. 2023 Jul 1;37(13-14):555-569. doi: 10.1101/gad.350479.123. Epub 2023 Jul 26. Genes Dev. 2023. PMID: 37495394 Free PMC article. Review.
-
Recent advancements in the structural biology of human telomerase and their implications for improved design of cancer therapeutics.NAR Cancer. 2023 Mar 3;5(1):zcad010. doi: 10.1093/narcan/zcad010. eCollection 2023 Mar. NAR Cancer. 2023. PMID: 36879683 Free PMC article.
-
POT1 recruits and regulates CST-Polα/Primase at human telomeres.bioRxiv [Preprint]. 2023 Oct 26:2023.05.08.539880. doi: 10.1101/2023.05.08.539880. bioRxiv. 2023. Update in: Cell. 2024 Jul 11;187(14):3638-3651.e18. doi: 10.1016/j.cell.2024.05.002. PMID: 37215005 Free PMC article. Updated. Preprint.
-
A persistent variant telomere sequence in a human pedigree.Nat Commun. 2024 Jun 1;15(1):4681. doi: 10.1038/s41467-024-49072-9. Nat Commun. 2024. PMID: 38824190 Free PMC article.
-
Structural biology of shelterin and telomeric chromatin: the pieces and an unfinished puzzle.Biochem Soc Trans. 2024 Aug 28;52(4):1551-1564. doi: 10.1042/BST20230300. Biochem Soc Trans. 2024. PMID: 39109533 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

