Mitochondrial homeostasis regulates definitive endoderm differentiation of human pluripotent stem cells
- PMID: 35177589
- PMCID: PMC8854419
- DOI: 10.1038/s41420-022-00867-z
Mitochondrial homeostasis regulates definitive endoderm differentiation of human pluripotent stem cells
Abstract
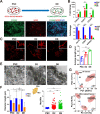
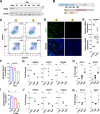
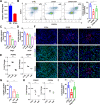
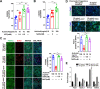
Cellular organelles play fundamental roles in almost all cell behaviors. Mitochondria have been reported to be functionally linked to various biological processes, including reprogramming and pluripotency maintenance. However, very little about the role of mitochondria has been revealed in human early development and lineage specification. Here, we reported the characteristics and function of mitochondria during human definitive endoderm differentiation. Using a well-established differentiation system, we first investigated the change of mitochondrial morphology by comparing undifferentiated pluripotent stem cells, the intermediate mesendoderm cells, and differentiated endoderm cells, and found that mitochondria were gradually elongated and matured along differentiation. We further analyzed the expression pattern of mitochondria-related genes by RNA-seq, indicating that mitochondria became active during differentiation. Supporting this notion, the production of adenosine triphosphate (ATP) and reactive oxygen species (ROS) was increased as well. Functionally, we utilized chemicals and genome editing techniques, which could interfere with mitochondrial homeostasis, to determine the role of mitochondria in human endoderm differentiation. Treatment with mitochondrial inhibitors, or genetic depletion of mitochondrial transcription factor A (TFAM), significantly reduced the differentiation efficiency of definitive endoderm. In addition, the defect in endoderm differentiation due to dysfunctional mitochondria could be restored to some extent by the addition of ATP. Moreover, the clearance of excessive ROS due to dysfunctional mitochondria by N-acetylcysteine (NAC) improved the differentiation as well. We further found that ATP and NAC could partially replace the growth factor activin A for definitive endoderm differentiation. Our study illustrates the essential role of mitochondria during human endoderm differentiation through providing ATP and regulating ROS levels, which may provide new insight for metabolic regulation of cell fate determination.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Khacho M, Harris R, Slack RS. Mitochondria as central regulators of neural stem cell fate and cognitive function. Nat Rev Neurosci. 2019;20:34–48. - PubMed
-
- Sun X, St John JC. The role of the mtDNA set point in differentiation, development and tumorigenesis. Biochem J. 2016;473:2955–71. - PubMed
-
- Gaspar JA, Doss MX, Hengstler JG, Cadenas C, Hescheler J, Sachinidis A. Unique metabolic features of stem cells, cardiomyocytes, and their progenitors. Circ Res. 2014;114:1346–60. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

