Late-Stage Chemoenzymatic Installation of Hydroxy-Bearing Allyl Moiety on the Indole Ring of Tryptophan-Containing Peptides
- PMID: 35178791
- PMCID: PMC9314954
- DOI: 10.1002/chem.202104614
Late-Stage Chemoenzymatic Installation of Hydroxy-Bearing Allyl Moiety on the Indole Ring of Tryptophan-Containing Peptides
Abstract
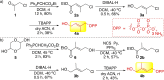
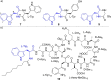
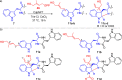
The late-stage functionalization of indole- and tryptophan-containing compounds with reactive moieties facilitates downstream diversification and leads to changes in their biological properties. Here, the synthesis of two hydroxy-bearing allyl pyrophosphates is described. A chemoenzymatic method is demonstrated which uses a promiscuous indole prenyltransferase enzyme to install a dual reactive hydroxy-bearing allyl moiety directly on the indole ring of tryptophan-containing peptides. This is the first report of late-stage indole modifications with this reactive group.
Keywords: C−H activation; allylation; biocatalysis; functionalization; indole prenyltransferase.
© 2022 The Authors. Chemistry - A European Journal published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

