Artificial intelligence in mammographic phenotyping of breast cancer risk: a narrative review
- PMID: 35184757
- PMCID: PMC8859891
- DOI: 10.1186/s13058-022-01509-z
Artificial intelligence in mammographic phenotyping of breast cancer risk: a narrative review
Abstract
Background: Improved breast cancer risk assessment models are needed to enable personalized screening strategies that achieve better harm-to-benefit ratio based on earlier detection and better breast cancer outcomes than existing screening guidelines. Computational mammographic phenotypes have demonstrated a promising role in breast cancer risk prediction. With the recent exponential growth of computational efficiency, the artificial intelligence (AI) revolution, driven by the introduction of deep learning, has expanded the utility of imaging in predictive models. Consequently, AI-based imaging-derived data has led to some of the most promising tools for precision breast cancer screening.
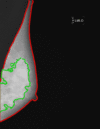
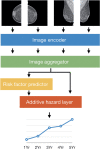
Main body: This review aims to synthesize the current state-of-the-art applications of AI in mammographic phenotyping of breast cancer risk. We discuss the fundamentals of AI and explore the computing advancements that have made AI-based image analysis essential in refining breast cancer risk assessment. Specifically, we discuss the use of data derived from digital mammography as well as digital breast tomosynthesis. Different aspects of breast cancer risk assessment are targeted including (a) robust and reproducible evaluations of breast density, a well-established breast cancer risk factor, (b) assessment of a woman's inherent breast cancer risk, and (c) identification of women who are likely to be diagnosed with breast cancers after a negative or routine screen due to masking or the rapid and aggressive growth of a tumor. Lastly, we discuss AI challenges unique to the computational analysis of mammographic imaging as well as future directions for this promising research field.
Conclusions: We provide a useful reference for AI researchers investigating image-based breast cancer risk assessment while indicating key priorities and challenges that, if properly addressed, could accelerate the implementation of AI-assisted risk stratification to future refine and individualize breast cancer screening strategies.
Keywords: Artificial intelligence; Breast cancer; Breast cancer risk; Breast density; Breast tomosynthesis; Deep learning; Digital mammography; Mammographic density; Mammographic imaging.
© 2022. The Author(s).
Conflict of interest statement
Dr. Conant reports membership on the Hologic, Inc. and iCAD, Inc., Scientific Advisory Boards and research grants with the same vendors; the other authors have no relevant conflicts of interest to disclose. The vendors had no role in the design of the review or the writing of the manuscript.
Figures


References
-
- Pace LE, Keating NL. A systematic assessment of benefits and risks to guide breast cancer screening decisions. JAMA. 2014;311(13):1327–1335. - PubMed
-
- McDonald ES, Clark AS, Tchou J, Zhang P, Freedman GM. Clinical diagnosis and management of breast cancer. J Nucl Med. 2016;57(Supplement 1):9S–16S. - PubMed
-
- Destounis SV, Santacroce A, Arieno A. Update on breast density, risk estimation, and supplemental screening. Am J Roentgenol. 2020;214(2):296–305. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

