Dopamine D1/D5 Receptor Signaling Is Involved in Arrhythmogenesis in the Setting of Takotsubo Cardiomyopathy
- PMID: 35187102
- PMCID: PMC8855058
- DOI: 10.3389/fcvm.2021.777463
Dopamine D1/D5 Receptor Signaling Is Involved in Arrhythmogenesis in the Setting of Takotsubo Cardiomyopathy
Abstract
Background: Previous studies suggested involvement of non-ß-adrenoceptors in the pathogenesis of Takotsubo cardiomyopathy (TTC). This study was designed to explore possible roles and underlying mechanisms of dopamine D1/D5 receptor coupled signaling in arrhythmogenesis of TTC.
Methods: Human-induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) were challenged by toxic concentration of epinephrine (Epi, 0.5 mM for 1 h) for mimicking the catecholamine excess in setting of TTC. Specific receptor blockers and activators were used to unveil roles of D1/D5 receptors. Patch clamp, qPCR, and FACS analyses were performed in the study.
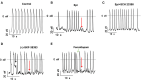
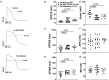
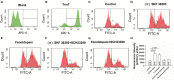
Results: High concentration Epi and two dopamine D1/D5 receptor agonists [(±)-SKF 38393 and fenoldopam] reduced the depolarization velocity and prolonged the duration of action potentials (APs) and caused arrhythmic events in iPSC-CMs, suggesting involvement of dopamine D1/D5 receptor signaling in arrhythmogenesis associated with QT interval prolongation in the setting of TTC. (±)-SKF 38393 and fenoldopam enhanced the reactive oxygen species (ROS)-production. H2O2 (100 μM) recapitulated the effects of (±)-SKF 38393 and fenoldopam on APs and a ROS-blocker N-acetylcysteine (NAC, 1 mM) abolished the effects, suggesting that the ROS-signaling is involved in the dopamine D1/D5 receptor actions. A NADPH oxidases blocker and a PKA- or PKC-blocker suppressed the effects of the dopamine receptor agonist, implying that PKA, NADPH oxidases and PKC participated in dopamine D1/D5 receptor signaling. The abnormal APs resulted from dopamine D1/D5 receptor activation-induced dysfunctions of ion channels including the Na+ and L-type Ca2+ and IKr channels.
Conclusions: Dopamine D1/D5 receptor signaling plays important roles for arrhythmogenesis of TTC. Dopamine D1/D5 receptor signaling in cardiomyocytes might be a potential target for treating arrhythmias in patients with TTC.
Keywords: D1/D5 dopamine receptor; Takotsubo cardiomyopathy; arrhythmia; catecholamine excess; human-induced pluripotent stem cell-derived cardiomyocytes.
Copyright © 2022 Huang, Yang, Li, Lan, Cyganek, Yuecel, Lang, Bieback, El-Battrawy, Zhou, Borggrefe and Akin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Roles and Mechanisms of Dopamine Receptor Signaling in Catecholamine Excess Induced Endothelial Dysfunctions.Int J Med Sci. 2024 Jul 22;21(10):1964-1975. doi: 10.7150/ijms.96550. eCollection 2024. Int J Med Sci. 2024. PMID: 39113882 Free PMC article.
-
Alpha 1-adrenoceptor signalling contributes to toxic effects of catecholamine on electrical properties in cardiomyocytes.Europace. 2021 Jul 18;23(7):1137-1148. doi: 10.1093/europace/euab008. Europace. 2021. PMID: 33604602
-
Exosomal mir-126-3p derived from endothelial cells induces ion channel dysfunction by targeting RGS3 signaling in cardiomyocytes: a novel mechanism in Takotsubo cardiomyopathy.Stem Cell Res Ther. 2025 Feb 4;16(1):36. doi: 10.1186/s13287-025-04157-0. Stem Cell Res Ther. 2025. PMID: 39901299 Free PMC article.
-
Coupling of D1 and D5 dopamine receptors to multiple G proteins: Implications for understanding the diversity in receptor-G protein coupling.Mol Neurobiol. 1998 Apr;16(2):125-34. doi: 10.1007/BF02740640. Mol Neurobiol. 1998. PMID: 9588624 Review.
-
Anti-atherosclerotic action of vascular D1 receptors.Clin Exp Pharmacol Physiol Suppl. 1999 Apr;26:S36-40. Clin Exp Pharmacol Physiol Suppl. 1999. PMID: 10386252 Review.
Cited by
-
Novel role of GRK2 in isoprenaline-induced activation of Na+/H+ exchanger 3 independent of β2-adrenergic receptor signaling.Mol Biol Rep. 2025 Feb 11;52(1):218. doi: 10.1007/s11033-025-10326-4. Mol Biol Rep. 2025. PMID: 39932653
-
Roles and Mechanisms of Dopamine Receptor Signaling in Catecholamine Excess Induced Endothelial Dysfunctions.Int J Med Sci. 2024 Jul 22;21(10):1964-1975. doi: 10.7150/ijms.96550. eCollection 2024. Int J Med Sci. 2024. PMID: 39113882 Free PMC article.
-
Role of Dopamine in the Heart in Health and Disease.Int J Mol Sci. 2023 Mar 6;24(5):5042. doi: 10.3390/ijms24055042. Int J Mol Sci. 2023. PMID: 36902474 Free PMC article. Review.
-
Risk Factors for Cardiac Complications in Patients With Pheochromocytoma and Paraganglioma: A Retrospective Single-Center Study.Front Endocrinol (Lausanne). 2022 Jun 1;13:877341. doi: 10.3389/fendo.2022.877341. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35721724 Free PMC article.
-
Contractile effects of stimulation of D1-dopamine receptors in the isolated human atrium.Naunyn Schmiedebergs Arch Pharmacol. 2025 Feb;398(2):1497-1508. doi: 10.1007/s00210-024-03340-z. Epub 2024 Aug 5. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39102031 Free PMC article.
References
-
- Dote K, Sato H, Tateishi H, Uchida T, Ishihara M. Myocardial stunning due to simultaneous multivessel coronary spasms: a review of 5 cases. J Cardiol. (1991) 21:203–14. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

