Ocimum sanctum, Zingiber officinale, and Piper nigrum extracts and their effects on gut microbiota modulations (prebiotic potential), basal inflammatory markers and lipid levels: oral supplementation study in healthy rats
- PMID: 35188051
- PMCID: PMC8865113
- DOI: 10.1080/13880209.2022.2033797
Ocimum sanctum, Zingiber officinale, and Piper nigrum extracts and their effects on gut microbiota modulations (prebiotic potential), basal inflammatory markers and lipid levels: oral supplementation study in healthy rats
Abstract
Context: Ocimum sanctum Linn (Labiatae) (OS), Zingiber officinale Rose (Zingiberaceae) (ZO), and Piper nigrum Linn (Piperaceae) (PN) are used in traditional medicine as immunomodulator, anti-inflammatory, and bioavailability enhancer agents.
Objective: Active phytoconstituents of OS, ZO, PN hydro-alcoholic extracts and their effects on gut microbiota, basal inflammation and lipid profile were investigated in rats.
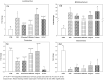
Materials and methods: Active phytoconstituents of extracts were analysed using HPLC and GC-MS. SD rats were supplemented with individual/combined extracts (OS-850; ZO-500; PN-100 mg/kg Bw) and Fructooligosaccharide (standard prebiotic-5g/kg-Bw), orally for 30 days. Haematology, lipid profile, LPS, CRP, IL-6, insulin and histology of vital organs were analysed. Caecal bacterial levels were assessed by RT-PCR.
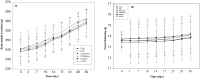
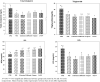
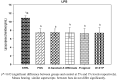
Results: High content of phenolic compounds luteolin-7-O-glucoside (430 ± 2.3 mg/100g), gallic acid (84.13 ± 1.2 mg/100 g) and flavones (88.18 ± 1.8 mg/100 g) were found in OS, ZO, and PN, respectively. Combined extract was rich in luteolin-7-O-glucoside (266.0 ± 1.80 mg/100 g). Essential oils including methyleugenol (13.96%), 6-shogaol (11.00%), piperine (18.26%), and cyclopentasiloxane (10.06%) were higher in OS, ZO, PN and combined extract. Higher levels of caecal Lactobacillus (1.7-3.4-fold), Bifidobacterium (5.89-28.4-fold), and lower levels of Firmicutes (0.04-0.91-fold), Bacteroides (0.69-0.88-fold) were noted among extracts and FOS supplemented rats. Significant (p < 0.05) decrease in plasma lipid profile and LPS was noted in all supplemented rats.
Discussion and conclusions: The current study could be first of its kind in exploring prebiotic potential of OS, ZO, PN and their effect on native gut bacterial population.
Keywords: Herbal extracts on endotoxin; beneficial bacteria; essential oils; phenolic compounds; systemic inflammation.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures




Similar articles
-
Effects of Piper nigrum extracts: Restorative perspectives of high-fat diet-induced changes on lipid profile, body composition, and hormones in Sprague-Dawley rats.Pharm Biol. 2015;53(9):1318-28. doi: 10.3109/13880209.2014.980585. Epub 2015 Apr 9. Pharm Biol. 2015. PMID: 25856709
-
In vitro assessment of the acaricidal activity of Piper longum, Piper nigrum, and Zingiber officinale extracts against Hyalomma anatolicum ticks.Exp Appl Acarol. 2017 Mar;71(3):303-317. doi: 10.1007/s10493-017-0113-2. Epub 2017 Mar 1. Exp Appl Acarol. 2017. PMID: 28251408
-
Attenuation of liver pro-inflammatory responses by Zingiber officinale via inhibition of NF-kappa B activation in high-fat diet-fed rats.Basic Clin Pharmacol Toxicol. 2012 Mar;110(3):238-44. doi: 10.1111/j.1742-7843.2011.00791.x. Epub 2011 Oct 21. Basic Clin Pharmacol Toxicol. 2012. PMID: 21902812
-
Anti-Inflammatory, gastrointestinal and hepatoprotective effects of Ocimum sanctum Linn: an ancient remedy with new application.Inflamm Allergy Drug Targets. 2013 Dec;12(6):378-84. doi: 10.2174/1871528112666131125110017. Inflamm Allergy Drug Targets. 2013. PMID: 24266685 Review.
-
A systematic review on black pepper (Piper nigrum L.): from folk uses to pharmacological applications.Crit Rev Food Sci Nutr. 2019;59(sup1):S210-S243. doi: 10.1080/10408398.2019.1565489. Epub 2019 Feb 11. Crit Rev Food Sci Nutr. 2019. PMID: 30740986
Cited by
-
Qualitative Profiling and Quantitative Analysis of Major Constituents in Jinmu-tang by UHPLC-Q-Orbitrap-MS and UPLC-TQ-MS/MS.Molecules. 2022 Nov 15;27(22):7887. doi: 10.3390/molecules27227887. Molecules. 2022. PMID: 36432001 Free PMC article.
-
Relevance of Indian Traditional Herbal Brews for Gut Microbiota Balance.Indian J Microbiol. 2024 Dec;64(4):1425-1444. doi: 10.1007/s12088-024-01251-4. Epub 2024 Mar 27. Indian J Microbiol. 2024. PMID: 39678955 Review.
-
HS-GC-IMS Analysis of Volatile Organic Compounds in Six Spicy Spices and Their Effects on Ulcerative Colitis.Molecules. 2024 Aug 8;29(16):3764. doi: 10.3390/molecules29163764. Molecules. 2024. PMID: 39202844 Free PMC article.
-
Effects of Luteolin-7-O-Glucoside on Intestinal Microbiota Dysbiosis and Drug Resistance Transmission Caused by Raoultella ornithinolytica B1645-1: Modulating the Composition of Intestinal Microbiota and Promoting the Transfer of blaNDM-1 Gene from Genus Enterococcus to Lactobacillus in Mice.Microorganisms. 2023 Oct 2;11(10):2477. doi: 10.3390/microorganisms11102477. Microorganisms. 2023. PMID: 37894135 Free PMC article.
-
Immunomodulatory Effect of Phytoactive Compounds on Human Health: A Narrative Review Integrated with Bioinformatics Approach.Curr Top Med Chem. 2024;24(12):1075-1100. doi: 10.2174/0115680266274272240321065039. Curr Top Med Chem. 2024. PMID: 38551050 Review.
References
-
- Ahmed HH, Salem AM, Sabry GM, Husein AA, Kotob SE.. 2013. Possible therapeutic uses of Salvia triloba and Piper nigrum in Alzheimer’s disease-induced rats. J Med Food. 16(5):437–446. - PubMed
-
- Alizadeh-Navaei R, Roozbeh F, Saravi M, Pouramir M, Jalali F, Moghadamnia AA.. 2008. Investigation of the effect of ginger on the lipid levels: a double blind controlled clinical trial. Saudi Med J. 29:1280–1284. - PubMed
-
- Babu KN, Hemalatha R, Satyanarayana U, Shujauddin M, Himaja N, Bhaskarachary K, Kumar BD.. 2018. Phytochemicals, polyphenols, prebiotic effect of Ocimum sanctum, Zingiber officinale, Piper nigrum extracts. J Herb Med. 13:42–51.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
