Metabolic Diversity and Aero-Tolerance in Anammox Bacteria from Geochemically Distinct Aquifers
- PMID: 35191775
- PMCID: PMC8862662
- DOI: 10.1128/msystems.01255-21
Metabolic Diversity and Aero-Tolerance in Anammox Bacteria from Geochemically Distinct Aquifers
Abstract
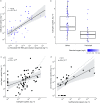
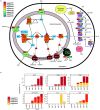
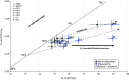
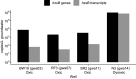
Anaerobic ammonium oxidation (anammox) is important for converting bioavailable nitrogen into dinitrogen gas, particularly in carbon-poor environments. However, the diversity and prevalence of anammox bacteria in the terrestrial subsurface-a typically oligotrophic environment-are little understood. To determine the distribution and activity of anammox bacteria across a range of aquifer lithologies and physicochemistries, we analyzed 16S rRNA genes and quantified hydrazine synthase genes and transcripts sampled from 59 groundwater wells and metagenomes and metatranscriptomes from an oxic-to-dysoxic subset. Data indicate that anammox and anammox-associated bacteria (class "Candidatus Brocadiae") are prevalent in the aquifers studied, and that anammox community composition is strongly differentiated by dissolved oxygen (DO), but not ammonia/nitrite. While "Candidatus Brocadiae" diversity decreased with increasing DO, "Candidatus Brocadiae" 16S rRNA genes and hydrazine synthase (hzsB) genes and transcripts were detected across a wide range of bulk groundwater DO concentrations (0 to 10 mg/L). Anammox genes and transcripts correlated significantly with those involved in aerobic ammonia oxidation (amoA), potentially representing a major source of nitrite for anammox. Eight "Candidatus Brocadiae" genomes (63 to 95% complete), representing 2 uncharacterized families and 6 novel species, were reconstructed. Six genomes have genes characteristic of anammox, all for chemolithoautotrophy. Anammox and aerotolerance genes of up to four "Candidatus Brocadiae" genomes were transcriptionally active under oxic and dysoxic conditions, although activity was highest in dysoxic groundwater. The coexpression of nrfAH nitrite reductase genes by "Candidatus Brocadiae" suggests active regeneration of ammonia for anammox. Our findings indicate that anammox bacteria contribute to loss of fixed N across diverse anoxic-to-oxic aquifer conditions, which is likely supported by nitrite from aerobic ammonia oxidation. IMPORTANCE Anammox is increasingly shown to play a major role in the aquatic nitrogen cycle and can outcompete heterotrophic denitrification in environments low in organic carbon. Given that aquifers are characteristically oligotrophic, anammox may represent a major route for the removal of fixed nitrogen in these environments, including agricultural nitrogen, a common groundwater contaminant. Our research confirms that anammox bacteria and the anammox process are prevalent in aquifers and occur across diverse lithologies (e.g., sandy gravel, sand-silt, and volcanic) and groundwater physicochemistries (e.g., various oxygen, carbon, nitrate, and ammonium concentrations). Results reveal niche differentiation among anammox bacteria largely driven by groundwater oxygen contents and provide evidence that anammox is supported by proximity to oxic niches and handoffs from aerobic ammonia oxidizers. We further show that this process, while anaerobic, is active in groundwater characterized as oxic, likely due to the availability of anoxic niches.
Keywords: aero-tolerance; ammonia oxidizers; anammox; aquifer; groundwater; “Candidatus Brocadiae”.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Nitrogen cycling and microbial cooperation in the terrestrial subsurface.ISME J. 2022 Nov;16(11):2561-2573. doi: 10.1038/s41396-022-01300-0. Epub 2022 Aug 8. ISME J. 2022. PMID: 35941171 Free PMC article.
-
"Candidatus Subterrananammoxibiaceae," a New Anammox Bacterial Family in Globally Distributed Marine and Terrestrial Subsurfaces.Appl Environ Microbiol. 2023 Aug 30;89(8):e0080023. doi: 10.1128/aem.00800-23. Epub 2023 Jul 20. Appl Environ Microbiol. 2023. PMID: 37470485 Free PMC article.
-
Role of Anaerobic Ammonium Oxidation (Anammox) in Nitrogen Removal from a Freshwater Aquifer.Environ Sci Technol. 2015 Oct 20;49(20):12169-77. doi: 10.1021/acs.est.5b02488. Epub 2015 Oct 1. Environ Sci Technol. 2015. PMID: 26401911
-
Anammox--growth physiology, cell biology, and metabolism.Adv Microb Physiol. 2012;60:211-62. doi: 10.1016/B978-0-12-398264-3.00003-6. Adv Microb Physiol. 2012. PMID: 22633060 Review.
-
Ecological characteristics of anaerobic ammonia oxidizing bacteria.Appl Microbiol Biotechnol. 2013 Mar;97(5):1841-9. doi: 10.1007/s00253-013-4697-0. Epub 2013 Jan 29. Appl Microbiol Biotechnol. 2013. PMID: 23358997 Review.
Cited by
-
Co-Occurrence and Cooperation between Comammox and Anammox Bacteria in a Full-Scale Attached Growth Municipal Wastewater Treatment Process.Environ Sci Technol. 2023 Mar 28;57(12):5013-5023. doi: 10.1021/acs.est.2c09223. Epub 2023 Mar 13. Environ Sci Technol. 2023. PMID: 36913533 Free PMC article.
-
Nitrogen fixation and diazotroph diversity in groundwater systems.ISME J. 2023 Nov;17(11):2023-2034. doi: 10.1038/s41396-023-01513-x. Epub 2023 Sep 15. ISME J. 2023. PMID: 37715043 Free PMC article.
-
Metabolic and phylogenetic diversity in the phylum Nitrospinota revealed by comparative genome analyses.ISME Commun. 2024 Jan 10;4(1):ycad017. doi: 10.1093/ismeco/ycad017. eCollection 2024 Jan. ISME Commun. 2024. PMID: 38317822 Free PMC article.
-
Community- and genome-based evidence for a shaping influence of redox potential on bacterial protein evolution.mSystems. 2023 Jun 29;8(3):e0001423. doi: 10.1128/msystems.00014-23. Epub 2023 Jun 8. mSystems. 2023. PMID: 37289197 Free PMC article.
-
Novel and unusual genes for nitrogen and metal cycling in Planctomycetota- and KSB1-affiliated metagenome-assembled genomes reconstructed from a marine subsea tunnel.FEMS Microbiol Lett. 2023 Jan 17;370:fnad049. doi: 10.1093/femsle/fnad049. FEMS Microbiol Lett. 2023. PMID: 37291701 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

