High-Throughput Precision Phenotyping of Left Ventricular Hypertrophy With Cardiovascular Deep Learning
- PMID: 35195663
- PMCID: PMC9008505
- DOI: 10.1001/jamacardio.2021.6059
High-Throughput Precision Phenotyping of Left Ventricular Hypertrophy With Cardiovascular Deep Learning
Abstract
Importance: Early detection and characterization of increased left ventricular (LV) wall thickness can markedly impact patient care but is limited by under-recognition of hypertrophy, measurement error and variability, and difficulty differentiating causes of increased wall thickness, such as hypertrophy, cardiomyopathy, and cardiac amyloidosis.
Objective: To assess the accuracy of a deep learning workflow in quantifying ventricular hypertrophy and predicting the cause of increased LV wall thickness.
Design, settings, and participants: This cohort study included physician-curated cohorts from the Stanford Amyloid Center and Cedars-Sinai Medical Center (CSMC) Advanced Heart Disease Clinic for cardiac amyloidosis and the Stanford Center for Inherited Cardiovascular Disease and the CSMC Hypertrophic Cardiomyopathy Clinic for hypertrophic cardiomyopathy from January 1, 2008, to December 31, 2020. The deep learning algorithm was trained and tested on retrospectively obtained independent echocardiogram videos from Stanford Healthcare, CSMC, and the Unity Imaging Collaborative.
Main outcomes and measures: The main outcome was the accuracy of the deep learning algorithm in measuring left ventricular dimensions and identifying patients with increased LV wall thickness diagnosed with hypertrophic cardiomyopathy and cardiac amyloidosis.
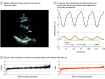
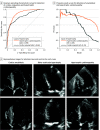
Results: The study included 23 745 patients: 12 001 from Stanford Health Care (6509 [54.2%] female; mean [SD] age, 61.6 [17.4] years) and 1309 from CSMC (808 [61.7%] female; mean [SD] age, 62.8 [17.2] years) with parasternal long-axis videos and 8084 from Stanford Health Care (4201 [54.0%] female; mean [SD] age, 69.1 [16.8] years) and 2351 from CSMS (6509 [54.2%] female; mean [SD] age, 69.6 [14.7] years) with apical 4-chamber videos. The deep learning algorithm accurately measured intraventricular wall thickness (mean absolute error [MAE], 1.2 mm; 95% CI, 1.1-1.3 mm), LV diameter (MAE, 2.4 mm; 95% CI, 2.2-2.6 mm), and posterior wall thickness (MAE, 1.4 mm; 95% CI, 1.2-1.5 mm) and classified cardiac amyloidosis (area under the curve [AUC], 0.83) and hypertrophic cardiomyopathy (AUC, 0.98) separately from other causes of LV hypertrophy. In external data sets from independent domestic and international health care systems, the deep learning algorithm accurately quantified ventricular parameters (domestic: R2, 0.96; international: R2, 0.90). For the domestic data set, the MAE was 1.7 mm (95% CI, 1.6-1.8 mm) for intraventricular septum thickness, 3.8 mm (95% CI, 3.5-4.0 mm) for LV internal dimension, and 1.8 mm (95% CI, 1.7-2.0 mm) for LV posterior wall thickness. For the international data set, the MAE was 1.7 mm (95% CI, 1.5-2.0 mm) for intraventricular septum thickness, 2.9 mm (95% CI, 2.4-3.3 mm) for LV internal dimension, and 2.3 mm (95% CI, 1.9-2.7 mm) for LV posterior wall thickness. The deep learning algorithm accurately detected cardiac amyloidosis (AUC, 0.79) and hypertrophic cardiomyopathy (AUC, 0.89) in the domestic external validation site.
Conclusions and relevance: In this cohort study, the deep learning model accurately identified subtle changes in LV wall geometric measurements and the causes of hypertrophy. Unlike with human experts, the deep learning workflow is fully automated, allowing for reproducible, precise measurements, and may provide a foundation for precision diagnosis of cardiac hypertrophy.
Conflict of interest statement
Figures

References
-
- National Human Genome Research Institute, National Institutes of Health. DNA sequencing costs: data. Accessed June 1, 2021. https://www.genome.gov/about-genomics/fact-sheets/DNA-Sequencing-Costs-Data
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

