Serially passaged, conditionally reprogrammed nasal epithelial cells as a model to study epithelial functions and SARS-CoV-2 infection
- PMID: 35196166
- PMCID: PMC8977148
- DOI: 10.1152/ajpcell.00363.2021
Serially passaged, conditionally reprogrammed nasal epithelial cells as a model to study epithelial functions and SARS-CoV-2 infection
Abstract
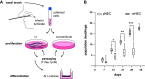
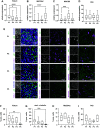
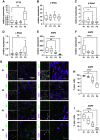
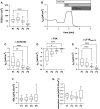
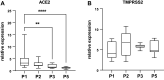
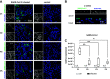
Primary airway epithelial cells (pAECs) cultivated at air-liquid interface (ALI) conditions are widely used as surrogates for human in vivo epithelia. To extend the proliferative capacity and to enable serially passaging of pAECs, conditional reprogramming (cr) has been employed in recent years. However, ALI epithelia derived from cr cells often display functional changes with increasing passages. This highlights the need for thorough validation of the ALI cultures for the respective application. In our study, we evaluated the use of serially passaged cr nasal epithelial cells (crNECs) as a model to study SARS-CoV-2 infection and effects on ion and water transport. NECs were obtained from healthy individuals and cultivated as ALI epithelia derived from passages 1, 2, 3, and 5. We compared epithelial differentiation, ion and water transport, and infection with SARS-CoV-2 between passages. Our results show that epithelia maintained major differentiation characteristics and physiological ion and water transport properties through all passages. However, the frequency of ciliated cells, short circuit currents reflecting epithelial Na+ channel (ENaC) and cystic fibrosis transmembrane conductance regulator (CFTR) activity and expression of aquaporin 3 and 5 decreased gradually over passages. crNECs also expressed SARS-CoV-2 receptors angiotensin converting enzyme 2 (ACE2) and transmembrane serin2 protease 2 (TMPRSS2) across all passages and allowed SARS-CoV-2 replication in all passages. In summary, we provide evidence that passaged crNECs provide an appropriate model to study SARS-CoV-2 infection and also epithelial transport function when considering some limitations that we defined herein.
Keywords: SARS-CoV-2; airway epithelial culture; conditional reprogramming; nasal epithelial cells.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

