Evaluation of Flow Cytometry for Cell Count and Detection of Bacteria in Biological Fluids
- PMID: 35196801
- PMCID: PMC8865548
- DOI: 10.1128/spectrum.01830-21
Evaluation of Flow Cytometry for Cell Count and Detection of Bacteria in Biological Fluids
Abstract
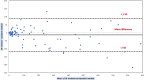
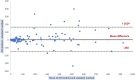
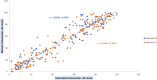
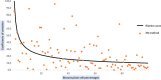
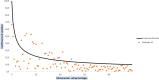
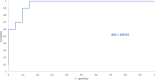
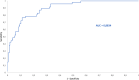
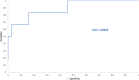
The analysis of biological fluids is crucial for the diagnosis and monitoring of diseases causing effusions and helps in the diagnosis of infectious diseases. The gold standard method for cell count in biological fluids is the manual method using counting chambers. The microbiological routine procedures consist of Direct Gram staining and culture on solid or liquid media. We evaluate the analytical performance of SYSMEX UF4000 (Sysmex, Kobe, Japan) and Sysmex XN10 (Sysmex, Kobe, Japan) in comparison with cytological and microbiological routine procedures. A total of 526 biological fluid samples were included in this study (42 ascitic, 31 pleural, 31 peritoneal, 125 cerebrospinal, 281 synovial, and 16 peritoneal dialysis fluids). All samples were analyzed by flow cytometry and subsequently processed following cytological and/or microbiological routine procedures. With regard to cell counts, UF4000 (Sysmex, Kobe, Japan) showed a performance that was at least equivalent to those of the reference methods and superior to those of XN10 (Sysmex, Kobe, Japan). Moreover, the bacterial count obtained with UF4000 (Sysmex, Kobe, Japan) was significantly higher among culture or Direct Gram stain positive samples. We established three optimal cutoff points to predict Direct Gram stain positive samples for peritoneal (465.0 bacteria/μL), synovial (1200.0 bacteria/μL), and cerebrospinal fluids (17.2 bacteria/μL) with maximum sensitivity and negative predictive values. Cell count and detection of bacteria by flow cytometry could be used upstream cytological and microbiological routine procedures to improve and accelerate the diagnosis of infection of biological fluid samples. IMPORTANCE The analysis of biological fluids is crucial for the diagnosis and monitoring of diseases causing effusions and helps in the diagnosis of infectious diseases. The possibility of carrying out cytological and microbiological analyses of biological fluid samples on the same automated machine would simplify the sample circuit (addressing the sample in a single laboratory, 24/7). It would also minimize the quantity of sample required. The performance of cytological and microbiological analysis by the flow cytometer UF 4000 (Sysmex, Kobe, Japan) has never been evaluated yet. This study has shown that bacterial count by flow cytometry using UF4000 (Sysmex, Kobe, Japan) could be used upstream of microbiological routine procedures to improve and to accelerate the diagnosis of infection of biological fluid samples.
Keywords: biological fluids; cell count; detection of bacteria; flow cytometry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Szamosi DI, Bautista JM, Cornbleet J, Glasser L, Rothe G, Sandhaus L, Key M, Meloni-Ehrig A, Culp NB, Dougherty W. 2006. Body fluids analysis for cellular composition: Approved Guidelines. Clinical Laboratory Standards Institute 26.
-
- Pushrich MA, Trzeciak S, Shapiro NI, Arnold RC, Horton JM, Studnek JR, Kline JA, Jones AE. 2011. Association between timing of antibiotics administration and mortality from septic shock in patients treated with a quantitative resuscitation protocol. Crit Care Med 39:2066–2071. doi: 10.1097/CCM.0b013e31821e87ab. - DOI - PMC - PubMed
-
- Garnacho-Montero J, Aldabo-Pallas T, Garnacho-Montero C, Cayuela A, Jiménez R, Barroso S, Ortiz-Leyba C. 2006. Timing of adequate antibiotic therapy is a greater determinant of outcome than are TNF and IL-10 polymorphisms in patients with sepsis. Critical Care 10:R111. doi: 10.1186/cc4995. - DOI - PMC - PubMed
-
- Buoro S, Ottomano C, Esposito SA, Gherardi P, Alessio MG, Crippa A, Raglio CF, Lippi G. 2013. Analytical and clinical evaluation of sysmex UF1000I for automated screening of cerebrospinal fluids. J Med Biochem 33:191–196. doi: 10.2478/jomb-2013-0034. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

