Recent improvements to the selectivity of extraction-based optical ion sensors
- PMID: 35201251
- PMCID: PMC8972301
- DOI: 10.1039/d1cc06636f
Recent improvements to the selectivity of extraction-based optical ion sensors
Abstract
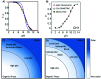
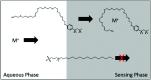
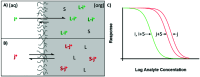
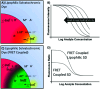
Optical sensors continue to demonstrate tremendous potential across a wide range of applications due to their high versatility and low cost. This feature article will focus on a number of recent advances made in improving the performance of extraction-based optical ion sensors within our group. This includes the progress of anchored solvatochromic transduction to provide pH and sample volume independent optical responses in nanoemulsion-based sensors. A recent breakthough is in polyion sensing in biological fluids that uses a novel indirect transduction mechanism that significantly improves the selectivity of dinonylnaphthalenesulfonate-based protamine sensors and its potential applications beyond polyion sensing. The role of particle stabilizers in relation to the response of emulsified sensors is shown to be important. Current challenges in the field and possible opportunities are also discussed.
Conflict of interest statement
There are no conflicts to declare.
Figures






Similar articles
-
Response Mechanism of Hyperpolarization-Based Polyion Nanosensors.ACS Sens. 2022 Oct 28;7(10):3108-3115. doi: 10.1021/acssensors.2c01599. Epub 2022 Sep 19. ACS Sens. 2022. PMID: 36121929
-
Response characteristics of a reversible electrochemical sensor for the polyion protamine.Anal Chem. 2005 Aug 15;77(16):5221-8. doi: 10.1021/ac050101l. Anal Chem. 2005. PMID: 16097762
-
Protamine/heparin optical nanosensors based on solvatochromism.Chem Sci. 2021 Nov 15;12(47):15596-15602. doi: 10.1039/d1sc04930e. eCollection 2021 Dec 8. Chem Sci. 2021. PMID: 35003589 Free PMC article.
-
Renovating the chromoionophores and detection modes in carrier-based ion-selective optical sensors.Anal Bioanal Chem. 2016 Apr;408(11):2717-25. doi: 10.1007/s00216-016-9406-2. Epub 2016 Feb 27. Anal Bioanal Chem. 2016. PMID: 26922342 Review.
-
Surface Plasmonic Sensors: Sensing Mechanism and Recent Applications.Sensors (Basel). 2021 Aug 4;21(16):5262. doi: 10.3390/s21165262. Sensors (Basel). 2021. PMID: 34450704 Free PMC article. Review.
Cited by
-
Perspective on fluorescence cell imaging with ionophore-based ion-selective nano-optodes.Biomicrofluidics. 2022 Jun 10;16(3):031301. doi: 10.1063/5.0090599. eCollection 2022 May. Biomicrofluidics. 2022. PMID: 35698631 Free PMC article.
-
Conducting polymer functionalization in search of advanced materials in ionometry: ion-selective electrodes and optodes.RSC Adv. 2024 Aug 13;14(35):25516-25548. doi: 10.1039/d4ra02615b. eCollection 2024 Aug 12. RSC Adv. 2024. PMID: 39139237 Free PMC article. Review.
-
Determination of Single-Ion Partition Coefficients between Water and Plasticized PVC Membrane Using Equilibrium-Based Techniques.Membranes (Basel). 2022 Oct 20;12(10):1019. doi: 10.3390/membranes12101019. Membranes (Basel). 2022. PMID: 36295778 Free PMC article.
-
GHz ultrasonic sensor for ionic content with high sensitivity and localization.iScience. 2023 May 19;26(6):106907. doi: 10.1016/j.isci.2023.106907. eCollection 2023 Jun 16. iScience. 2023. PMID: 37305695 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

