Chemical Synthesis of a Functional Fluorescent-Tagged α-Bungarotoxin
- PMID: 35202107
- PMCID: PMC8879871
- DOI: 10.3390/toxins14020079
Chemical Synthesis of a Functional Fluorescent-Tagged α-Bungarotoxin
Abstract
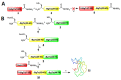
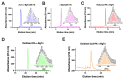
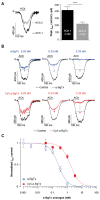
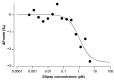
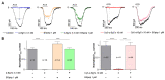
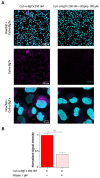
α-bungarotoxin is a large, 74 amino acid toxin containing five disulphide bridges, initially identified in the venom of Bungarus multicinctus snake. Like most large toxins, chemical synthesis of α-bungarotoxin is challenging, explaining why all previous reports use purified or recombinant α-bungarotoxin. However, only chemical synthesis allows easy insertion of non-natural amino acids or new chemical functionalities. Herein, we describe a procedure for the chemical synthesis of a fluorescent-tagged α-bungarotoxin. The full-length peptide was designed to include an alkyne function at the amino-terminus through the addition of a pentynoic acid linker. Chemical synthesis of α-bungarotoxin requires hydrazide-based coupling of three peptide fragments in successive steps. After completion of the oxidative folding, an azide-modified Cy5 fluorophore was coupled by click chemistry onto the toxin. Next, we determined the efficacy of the fluorescent-tagged α-bungarotoxin to block acetylcholine (ACh)-mediated currents in response to muscle nicotinic receptor activation in TE671 cells. Using automated patch-clamp recordings, we demonstrate that fluorescent synthetic α-bungarotoxin has the expected nanomolar affinity for the nicotinic receptor. The blocking effect of fluorescent α-bungarotoxin could be displaced by incubation with a 20-mer peptide mimicking the α-bungarotoxin binding site. In addition, TE671 cells could be labelled with fluorescent toxin, as witnessed by confocal microscopy, and this labelling was partially displaced by the 20-mer competitive peptide. We thus demonstrate that synthetic fluorescent-tagged α-bungarotoxin preserves excellent properties for binding onto muscle nicotinic receptors.
Keywords: TE671 cells; automated patch-clamp; click chemistry; fluorescent peptide; native chemical ligation; nicotinic acetylcholine receptor; peptide chemistry; toxins; α-bungarotoxin.
Conflict of interest statement
The authors Claude Zoukimian, Rémy Béroud and Michel De Waard declare the following competing interest: employee, CEO and founder of Smartox Biotechnology, respectively. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript or in the decision to publish the results.
Figures
Similar articles
-
Kappa 2-bungarotoxin and kappa 3-bungarotoxin: two new neuronal nicotinic receptor antagonists isolated from the venom of Bungarus multicinctus.Brain Res. 1990 Feb 19;509(2):237-48. doi: 10.1016/0006-8993(90)90548-p. Brain Res. 1990. PMID: 2322821
-
Identification of alpha-bungarotoxin (A31) as the major postsynaptic neurotoxin, and complete nucleotide identity of a genomic DNA of Bungarus candidus from Java with exons of the Bungarus multicinctus alpha-bungarotoxin (A31) gene.Toxicon. 2003 Sep 15;42(4):381-90. doi: 10.1016/s0041-0101(03)00168-5. Toxicon. 2003. PMID: 14505938
-
Galantamine activates muscle-type nicotinic acetylcholine receptors without binding to the acetylcholine-binding site.J Neurosci. 2005 Feb 23;25(8):1992-2001. doi: 10.1523/JNEUROSCI.4985-04.2005. J Neurosci. 2005. PMID: 15728839 Free PMC article.
-
Three-finger alpha-neurotoxins and the nicotinic acetylcholine receptor, forty years on.J Pharmacol Sci. 2004 Jan;94(1):1-17. doi: 10.1254/jphs.94.1. J Pharmacol Sci. 2004. PMID: 14745112 Review.
-
Thymopoietin, a thymic polypeptide, potently interacts at muscle and neuronal nicotinic alpha-bungarotoxin receptors.Mol Neurobiol. 1992 Spring;6(1):19-40. doi: 10.1007/BF02935565. Mol Neurobiol. 1992. Retraction in: Mol Neurobiol. 1993 Summer;7(2):160. doi: 10.1007/BF02935641. PMID: 1463587 Retracted. Review.
Cited by
-
Evaluation of [125I]α-Bungarotoxin Binding to α7 Nicotinic Acetylcholinergic Receptors in Hippocampus-Subiculum of Postmortem Human Alzheimer's Disease Brain.Receptors (Basel). 2025 Mar;4(1):7. doi: 10.3390/receptors4010007. Epub 2025 Mar 20. Receptors (Basel). 2025. PMID: 40678024 Free PMC article.
-
Editorial of the Special Issue "Toxins: Mr Hyde or Dr Jekyll?".Toxins (Basel). 2023 Feb 10;15(2):142. doi: 10.3390/toxins15020142. Toxins (Basel). 2023. PMID: 36828456 Free PMC article.
-
A Pore Forming Toxin-like Protein Derived from Chinese Red Belly Toad Bombina maxima Triggers the Pyroptosis of Hippomal Neural Cells and Impairs the Cognitive Ability of Mice.Toxins (Basel). 2023 Mar 3;15(3):191. doi: 10.3390/toxins15030191. Toxins (Basel). 2023. PMID: 36977082 Free PMC article.
-
Shining a Light on Venom-Peptide Receptors: Venom Peptides as Targeted Agents for In Vivo Molecular Imaging.Toxins (Basel). 2024 Jul 4;16(7):307. doi: 10.3390/toxins16070307. Toxins (Basel). 2024. PMID: 39057947 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

