From Light to Structure: Photo Initiators for Radical Two-Photon Polymerization
- PMID: 35202499
- PMCID: PMC9324900
- DOI: 10.1002/chem.202104191
From Light to Structure: Photo Initiators for Radical Two-Photon Polymerization
Abstract
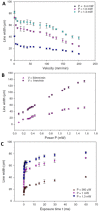
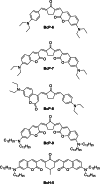
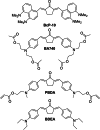
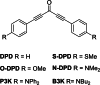
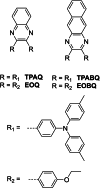
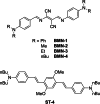
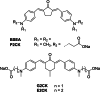
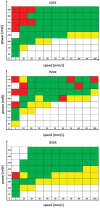
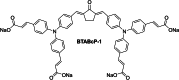
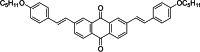
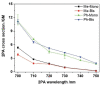
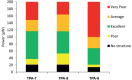
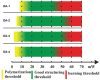
Two-photon polymerization (2PP) represents a powerful technique for the fabrication of precise three-dimensional structures on a micro- and nanometer scale for various applications. While many review articles are focusing on the used polymeric materials and their application in 2PP, in this review the class of two-photon photo initiators (2PI) used for radical polymerization is discussed in detail. Because the demand for highly efficient 2PI has increased in the last decades, different approaches in designing new efficient 2PIs occurred. This review summarizes the 2PIs known in literature and discusses their absorption behavior under one- and two-photon absorption (2PA) conditions, their two-photon cross sections (σTPA ) as well as their efficiency under 2PP conditions. Here, the photo initiators are grouped depending on their chromophore system (D-π-A-π-D, D-π-D, etc.). Their polymerization efficiencies are evaluated by fabrication windows (FW) depending on different laser intensities and writing speeds.
Keywords: microfabrication; photo initiator; two-photon absorption; two-photon cross-section; two-photon polymerization.
© 2022 The Authors. Chemistry - A European Journal published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







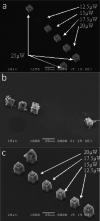

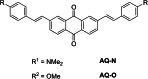
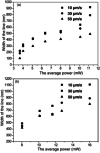
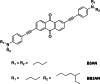






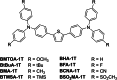




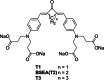



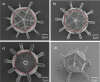
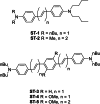

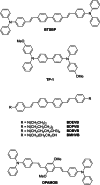

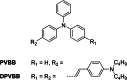

















Similar articles
-
Wavelength-optimized Two-Photon Polymerization Using Initiators Based on Multipolar Aminostyryl-1,3,5-triazines.Sci Rep. 2018 Nov 22;8(1):17273. doi: 10.1038/s41598-018-35301-x. Sci Rep. 2018. PMID: 30467346 Free PMC article.
-
Functional Materials for Two-Photon Polymerization in Microfabrication.Small. 2019 Oct;15(40):e1902687. doi: 10.1002/smll.201902687. Epub 2019 Aug 12. Small. 2019. PMID: 31402578 Review.
-
Lighting the Way to See Inside Two-Photon Absorption Materials: Structure-Property Relationship and Biological Imaging.Materials (Basel). 2017 Feb 23;10(3):223. doi: 10.3390/ma10030223. Materials (Basel). 2017. PMID: 28772584 Free PMC article. Review.
-
Beyond the Threshold: A Study of Chalcogenophene-Based Two-Photon Initiators.Chem Mater. 2022 Apr 12;34(7):3042-3052. doi: 10.1021/acs.chemmater.1c04002. Epub 2022 Mar 22. Chem Mater. 2022. PMID: 35431440 Free PMC article.
-
Four new two-photon polymerization initiators with varying donor and conjugated bridge: synthesis and two-photon activity.Spectrochim Acta A Mol Biomol Spectrosc. 2014 Jan 24;118:538-42. doi: 10.1016/j.saa.2013.09.051. Epub 2013 Sep 18. Spectrochim Acta A Mol Biomol Spectrosc. 2014. PMID: 24084482
Cited by
-
High- and Low-Fluorescent Photoinitiators for Multiphoton Lithography.ACS Appl Polym Mater. 2025 Jul 31;7(15):10108-10120. doi: 10.1021/acsapm.5c01802. eCollection 2025 Aug 8. ACS Appl Polym Mater. 2025. PMID: 40809104 Free PMC article.
-
Two-Photon Polymerization: Fundamentals, Materials, and Chemical Modification Strategies.Adv Sci (Weinh). 2023 Mar;10(7):e2204072. doi: 10.1002/advs.202204072. Epub 2022 Dec 30. Adv Sci (Weinh). 2023. PMID: 36585380 Free PMC article. Review.
-
Three-dimensional printing in modern orthopedic trauma surgery: a comprehensive analysis of technical evolution and clinical translation.Front Med (Lausanne). 2025 Jul 15;12:1560909. doi: 10.3389/fmed.2025.1560909. eCollection 2025. Front Med (Lausanne). 2025. PMID: 40735441 Free PMC article. Review.
-
Push-Pull Carbazole-Based Dyes: Synthesis, Strong Ultrafast Nonlinear Optical Response, and Effective Photoinitiation for Multiphoton Lithography.ACS Appl Opt Mater. 2024 Aug 5;2(8):1653-1666. doi: 10.1021/acsaom.4c00241. eCollection 2024 Aug 23. ACS Appl Opt Mater. 2024. PMID: 39206344 Free PMC article.
-
3D printing by stereolithography using thermal initiators.Nat Commun. 2024 Mar 13;15(1):2285. doi: 10.1038/s41467-024-46532-0. Nat Commun. 2024. PMID: 38480705 Free PMC article.
References
-
- Göppert-Mayer M., Ann. Phys. 1931, 401, 273–294.
-
- Kaiser W., Garrett C. G. B., Phys. Rev. Lett. 1961, 7, 229–231.
-
- Maruo S., Nakamura O., Kawata S., Opt. Lett. 1997, 22, 132–134. - PubMed
-
- Wollhofen R., Buchegger B., Eder C., Jacak J., Kreutzer J., Klar T. A., Opt. Mater. Express 2017, 7, 2538–2559.
-
- Claus T. K., Richter B., Hahn V., Welle A., Kayser S., Wegener M., Bastmeyer M., Delaittre G., Barner-Kowollik C., Angew. Chem. Int. Ed. 2016, 55, 3817–3822; - PubMed
- Angew. Chem. 2016, 128, 3882–3887.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

