Gut Microbiota-Derived Small Extracellular Vesicles Endorse Memory-like Inflammatory Responses in Murine Neutrophils
- PMID: 35203650
- PMCID: PMC8962420
- DOI: 10.3390/biomedicines10020442
Gut Microbiota-Derived Small Extracellular Vesicles Endorse Memory-like Inflammatory Responses in Murine Neutrophils
Abstract
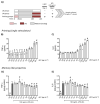
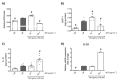
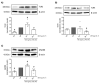
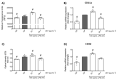
Neutrophils are classically characterized as merely reactive innate effector cells. However, the microbiome is known to shape the education and maturation process of neutrophils, improving their function and immune-plasticity. Recent reports demonstrate that murine neutrophils possess the ability to exert adaptive responses after exposure to bacterial components such as LPS (Gram-negative bacteria) or LTA (Gram-positive bacteria). We now ask whether small extracellular vesicles (EVs) from the gut may directly mediate adaptive responses in neutrophils in vitro. Murine bone marrow-derived neutrophils were primed in vitro by small EVs of high purity collected from colon stool samples, followed by a second hit with LPS. We found that low-dose priming with gut microbiota-derived small EVs enhanced pro-inflammatory sensitivity as indicated by elevated levels of TNF-α, IL-6, ROS and MCP-1 and increased migratory and phagocytic activity. In contrast, high-dose priming resulted in a tolerant phenotype, marked by increased IL-10 and decreased transmigration and phagocytosis. Alterations in TLR2/MyD88 as well as TLR4/MyD88 signaling were correlated with the induction of adaptive cues in neutrophils in vitro. Taken together, our study shows that small EVs from stools can drive adaptive responses in neutrophils in vitro and may represent a missing link in the gut-immune axis.
Keywords: inflammation; memory-like; microbiota; neutrophils; phagocytosis; priming; small EVs; transmigration.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

