Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy
- PMID: 35208478
- PMCID: PMC8877936
- DOI: 10.3390/medicina58020155
Clinical Value of EZH2 in Hepatocellular Carcinoma and Its Potential for Target Therapy
Abstract
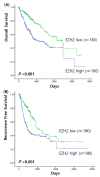
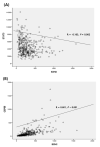
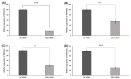
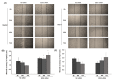
Background and objectives: EZH2 is overexpressed in hepatocellular carcinoma (HCC) and is correlated with poor prognosis. However, its clinical significance and molecular mechanism have not been studied in HCC. In this study, clinical and prognostic values of EZH2 was studied using Total Cancer Genome Atlas (TCGA) data and then, these data were confirmed in Huh1 and HepG2 cell lines. Materials and Methods: We used the TCGA database from cBioPortal. In addition, we analyzed EZH2 mRNA levels in HCC cell lines and its correlation with STAT3 and EZH2. Results: According to TCGA, EZH2 had a prognostic value in various cancers, especially in HCC. Furthermore, EZH2 in HCC was correlated with N stage (p = 0.045) and alpha-fetoprotein (AFP) > 20 ng/mL (p < 0.01). However, a negative association between EZH2 and age (p = 0.027) was found. The overall survival result of HCC was significantly poorer in patients with high EZH2 expression. In addition, the recurrence rate was also significantly higher in patients with high expression of EZH2 than those with low expression (χ2 = 16.10, p < 0.001). EZH2 expression was negatively correlated with STAT3 expression among EZH2-associated genes (R = -0.163, p = 0.002). EZH2 expression level was down-regulated to 50% or less compared to the control group treated negative siRNA. MTT assays showed that EZH2-siRNA affected on the viability of HCC cell line significantly. Conclusions: In conclusion, the overexpression of EZH2 was an independent biomarker for poor outcomes of HCC. However, more in vivo studies are required to identify the downstream target genes in HCC to improve our understanding of the biological role of EZH2 in HCC.
Keywords: EZH2; STAT3; TCGA; hepatocellular carcinoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

