Experimental Infection of Captive Red Foxes (Vulpes vulpes) with Mycobacterium bovis
- PMID: 35208835
- PMCID: PMC8879846
- DOI: 10.3390/microorganisms10020380
Experimental Infection of Captive Red Foxes (Vulpes vulpes) with Mycobacterium bovis
Abstract
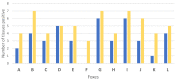
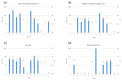
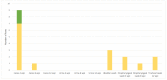
In Europe, animal tuberculosis (TB) due to Mycobacterium bovis involves multi-host communities that include cattle and wildlife species, such as wild boar (Sus scrofa), badgers (Meles meles) and red deer (Cervus elaphus). Red fox (Vulpes vulpes) infections have also been recently reported in some TB endemic regions in the Iberian Peninsula and France, with some of the infected animals shedding M. bovis in urine and feces. In order to understand the pathogenesis of M. bovis infection in foxes and the associated risk of transmission, 12 captive foxes (6 females and 6 males) were inoculated orally with 2 × 107 colony-forming units of a French field isolate of M. bovis. Clinical samples (urine, feces and oropharyngeal swabs) were collected every four weeks and tested for molecular diagnosis and bacteriology. Serological responses were measured by IDEXX M. bovis Ab Test and Multi Antigen Print Immunoassay (MAPIA). At a post-mortem examination performed 12 weeks post infection (wpi), tissues were tested for the presence of M. bovis and associated gross and microscopic TB-like lesions. M. bovis was detected by PCR in bladder swabs of 3 animals at 12 wpi. It was also detected pre-mortem at different time points of the experiment in the oropharyngeal mucus of three individuals and in the feces of nine foxes, with two of them confirmed by bacteriology. All 12 foxes had at least 4 PCR positive samples (out of the 23 tested), and all but 1 fox had at least 1 culture positive sample. The culture negative fox was PCR positive in both retropharyngeal and mesenteric lymph nodes, in line with the results of the other animals. Seroconversion was observed in all foxes except one during the experiment, and in nine at the final time point. No gross visible lesions were found in any animal at the post-mortem examination. The histology showed small granulomas within the lymph nodes, tonsils, liver and lungs from eight animals, with the presence of few acid-fast bacilli. These results confirmed that all orally-infected foxes developed mild TB lesions but they were able to shed mycobacteria in about 75% of cases, 1 month post-infection (9 out 12 foxes). These results show that it is possible to induce typical TB infection experimentally in captive foxes, with measurable M. bovis excretion; such an experimental system could be useful for future evaluations of diagnostics and vaccines in this species.
Keywords: Vulpes vulpes; excretion; pathogenesis; red fox; serology; tuberculosis.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures


References
-
- Santos N., Richomme C., Nunes T., Vicente J., Alves P.C., de la Fuente J., Correia-Neves M., Boschiroli M.-L., Delahay R., Gortázar C. Quantification of the Animal Tuberculosis Multi-Host Community Offers Insights for Control. Pathogens. 2020;9:421. doi: 10.3390/pathogens9060421. - DOI - PMC - PubMed
-
- Delahay R.J., Smith G.C., Barlow A.M., Walker N., Harris A., Clifton-Hadley R.S., Cheeseman C.L. Bovine Tuberculosis Infection in Wild Mammals in the South-West Region of England: A Survey of Prevalence and a Semi-Quantitative Assessment of the Relative Risks to Cattle. Vet. J. 2007;173:287–301. doi: 10.1016/j.tvjl.2005.11.011. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

