Endothelial Ion Channels and Cell-Cell Communication in the Microcirculation
- PMID: 35211031
- PMCID: PMC8861442
- DOI: 10.3389/fphys.2022.805149
Endothelial Ion Channels and Cell-Cell Communication in the Microcirculation
Abstract
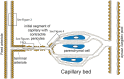
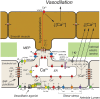
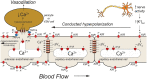
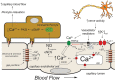
Endothelial cells in resistance arteries, arterioles, and capillaries express a diverse array of ion channels that contribute to Cell-Cell communication in the microcirculation. Endothelial cells are tightly electrically coupled to their neighboring endothelial cells by gap junctions allowing ion channel-induced changes in membrane potential to be conducted for considerable distances along the endothelial cell tube that lines arterioles and forms capillaries. In addition, endothelial cells may be electrically coupled to overlying smooth muscle cells in arterioles and to pericytes in capillaries via heterocellular gap junctions allowing electrical signals generated by endothelial cell ion channels to be transmitted to overlying mural cells to affect smooth muscle or pericyte contractile activity. Arteriolar endothelial cells express inositol 1,4,5 trisphosphate receptors (IP3Rs) and transient receptor vanilloid family member 4 (TRPV4) channels that contribute to agonist-induced endothelial Ca2+ signals. These Ca2+ signals then activate intermediate and small conductance Ca2+-activated K+ (IKCa and SKCa) channels causing vasodilator-induced endothelial hyperpolarization. This hyperpolarization can be conducted along the endothelium via homocellular gap junctions and transmitted to overlying smooth muscle cells through heterocellular gap junctions to control the activity of voltage-gated Ca2+ channels and smooth muscle or pericyte contraction. The IKCa- and SKCa-induced hyperpolarization may be amplified by activation of inward rectifier K+ (KIR) channels. Endothelial cell IP3R- and TRPV4-mediated Ca2+ signals also control the production of endothelial cell vasodilator autacoids, such as NO, PGI2, and epoxides of arachidonic acid contributing to control of overlying vascular smooth muscle contractile activity. Cerebral capillary endothelial cells lack IKCa and SKCa but express KIR channels, IP3R, TRPV4, and other Ca2+ permeable channels allowing capillary-to-arteriole signaling via hyperpolarization and Ca2+. This allows parenchymal cell signals to be detected in capillaries and signaled to upstream arterioles to control blood flow to capillaries by active parenchymal cells. Thus, endothelial cell ion channels importantly participate in several forms of Cell-Cell communication in the microcirculation that contribute to microcirculatory function and homeostasis.
Keywords: arterioles; blood flow; capillaries; endothelial cells; endothelial ion channels and cell-cell communication ion channels; functional hyperemia; pericytes; vascular smooth muscle cells.
Copyright © 2022 Jackson.
Conflict of interest statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Calcium-Dependent Ion Channels and the Regulation of Arteriolar Myogenic Tone.Front Physiol. 2021 Nov 8;12:770450. doi: 10.3389/fphys.2021.770450. eCollection 2021. Front Physiol. 2021. PMID: 34819877 Free PMC article. Review.
-
Potassium channels in the peripheral microcirculation.Microcirculation. 2005 Jan-Feb;12(1):113-27. doi: 10.1080/10739680590896072. Microcirculation. 2005. PMID: 15804979 Free PMC article. Review.
-
Endothelial potassium channels, endothelium-dependent hyperpolarization and the regulation of vascular tone in health and disease.Clin Exp Pharmacol Physiol. 2004 Sep;31(9):641-9. doi: 10.1111/j.1440-1681.2004.04053.x. Clin Exp Pharmacol Physiol. 2004. PMID: 15479173 Review.
-
Inward rectifier potassium (Kir2.1) channels as end-stage boosters of endothelium-dependent vasodilators.J Physiol. 2016 Jun 15;594(12):3271-85. doi: 10.1113/JP271652. Epub 2016 Mar 4. J Physiol. 2016. PMID: 26840527 Free PMC article.
-
Boosting the signal: Endothelial inward rectifier K+ channels.Microcirculation. 2017 Apr;24(3):10.1111/micc.12319. doi: 10.1111/micc.12319. Microcirculation. 2017. PMID: 27652592 Free PMC article. Review.
Cited by
-
Vasodilators mobilize SK3 channels in endothelial cells to produce arterial relaxation.Proc Natl Acad Sci U S A. 2023 Aug;120(31):e2303238120. doi: 10.1073/pnas.2303238120. Epub 2023 Jul 26. Proc Natl Acad Sci U S A. 2023. PMID: 37494394 Free PMC article.
-
Association between peripheral and coronary microvascular function and the impact of myocardial bridging.Physiol Rep. 2025 Apr;13(8):e70340. doi: 10.14814/phy2.70340. Physiol Rep. 2025. PMID: 40268885 Free PMC article.
-
Morphological and Functional Remodeling of Vascular Endothelium in Cardiovascular Diseases.Int J Mol Sci. 2023 Jan 19;24(3):1998. doi: 10.3390/ijms24031998. Int J Mol Sci. 2023. PMID: 36768314 Free PMC article. Review.
-
Cracking the Endothelial Calcium (Ca2+) Code: A Matter of Timing and Spacing.Int J Mol Sci. 2023 Nov 26;24(23):16765. doi: 10.3390/ijms242316765. Int J Mol Sci. 2023. PMID: 38069089 Free PMC article. Review.
-
Conditions for Kir-induced bistability of membrane potential in capillary endothelial cells.Math Biosci. 2023 Jan;355:108955. doi: 10.1016/j.mbs.2022.108955. Epub 2022 Dec 10. Math Biosci. 2023. PMID: 36513149 Free PMC article.
References
-
- Abdullaev I. F., Bisaillon J. M., Potier M., Gonzalez J. C., Motiani R. K., Trebak M. (2008). STIM1 and Orai1 mediate CRAC currents and store-operated calcium entry important for endothelial cell proliferation. Circ. Res. 103, 1289–1299. doi: 10.1161/01.res.0000338496.95579.56, PMID: - DOI - PMC - PubMed
-
- Bagher P., Beleznai T., Kansui Y., Mitchell R., Garland C. J., Dora K. A. (2012). Low intravascular pressure activates endothelial cell TRPV4 channels, local Ca2+ events, and IKCa channels, reducing arteriolar tone. Proc. Natl. Acad. Sci. U. S. A. 109, 18174–18179. doi: 10.1073/pnas.1211946109, PMID: - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

