Comparison of two staining techniques on the manual and automated canine sperm morphology analysis
- PMID: 35212033
- PMCID: PMC9311200
- DOI: 10.1111/rda.14100
Comparison of two staining techniques on the manual and automated canine sperm morphology analysis
Abstract
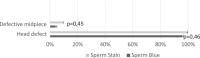
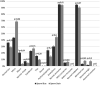
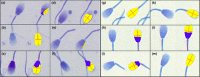
Detailed and direct analysis of semen, including sperm morphology, enables a diagnosis of male fertility. This study aimed to describe an economical and verified protocol for canine spermiograms and compare the effectiveness of Sperm Stain® and Sperm Blue® (Microptic, Spain) in veterinary practice. Sperm assessment was conducted manually, using a standard optical microscope, and via computerized semen analysis using the SCA® CASA (Sperm Class Analyzer® CASA System-MICROPTIC, Spain). This study showed that Sperm Blue® is a better solution for computerized sperm quality analysis of healthy dogs. At the same time, Sperm Stain® turned out to be more helpful in identifying specific morphological defects of sperm. Automated canine sperm morphology analysis worked better with Sperm Blue stain, but Sperm Stain simplified manual evaluation of various organelles' defects. Standard, manual examination is more error-prone for an inexperienced andrology technician, but it seems to be still a gold standard technique for canine sperm assessment.
Keywords: CASA; Sperm Blue®; Sperm Stain®; andrology; dog spermiogram; sperm morphology.
© 2022 The Authors. Reproduction in Domestic Animals published by Wiley-VCH GmbH.
Conflict of interest statement
The authors whose names are listed immediately below certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership or other equity interest; and expert testimony or patent‐licensing arrangements) or non‐financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Figures
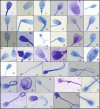

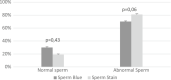
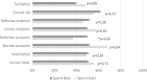
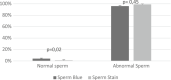
Similar articles
-
Dog sperm DNA: Raw semen evaluation with Toluidine blue stain.Reprod Domest Anim. 2019 Aug;54(8):1078-1084. doi: 10.1111/rda.13490. Epub 2019 Jun 26. Reprod Domest Anim. 2019. PMID: 31187909
-
Viability assessment of spermatozoa in large falcons (Falco spp.) using various staining protocols.Reprod Domest Anim. 2020 Oct;55(10):1383-1392. doi: 10.1111/rda.13785. Epub 2020 Aug 17. Reprod Domest Anim. 2020. PMID: 32722862
-
Manual vs. computer-assisted sperm analysis: can CASA replace manual assessment of human semen in clinical practice?Ginekol Pol. 2017;88(2):56-60. doi: 10.5603/GP.a2017.0012. Ginekol Pol. 2017. PMID: 28326513
-
Evaluation of Canine Sperm and Management of Semen Disorders.Vet Clin North Am Small Anim Pract. 2018 Jul;48(4):533-545. doi: 10.1016/j.cvsm.2018.02.003. Epub 2018 Apr 17. Vet Clin North Am Small Anim Pract. 2018. PMID: 29678334 Review.
-
CASA in the medical laboratory: CASA in diagnostic andrology and assisted conception.Reprod Fertil Dev. 2018 Jun;30(6):850-859. doi: 10.1071/RD17520. Reprod Fertil Dev. 2018. PMID: 29559071 Review.
Cited by
-
Influence of two collection frequency intervals on sperm quality of standard and miniature bull Terriers during short breeding periods: A clinical field study.Vet World. 2024 Apr;17(4):820-828. doi: 10.14202/vetworld.2024.820-828. Epub 2024 Apr 15. Vet World. 2024. PMID: 38798297 Free PMC article.
-
Standardisation of Dog Sperm Morphology Classification.Reprod Domest Anim. 2025 Feb;60(2):e70024. doi: 10.1111/rda.70024. Reprod Domest Anim. 2025. PMID: 39989363 Free PMC article.
-
Application of Staining Methods to Compare Chromatin Condensation in Fresh and Freeze-Thawed Dog Semen.Reprod Domest Anim. 2025 Mar;60(3):e70023. doi: 10.1111/rda.70023. Reprod Domest Anim. 2025. PMID: 40022439 Free PMC article.
-
Osteopontin Concentration in Prostates Fractions: A Novel Marker of Sperm Quality in Dogs.Vet Sci. 2023 Nov 8;10(11):646. doi: 10.3390/vetsci10110646. Vet Sci. 2023. PMID: 37999469 Free PMC article.
-
Improving the Diagnosis and Follow-Up of Chronic Myeloid Leukemia Using Conventional and Molecular Techniques.J Clin Lab Anal. 2025 Mar;39(5):e70001. doi: 10.1002/jcla.70001. Epub 2025 Feb 10. J Clin Lab Anal. 2025. PMID: 39927600 Free PMC article.
References
-
- Brito, M. M. , Angrimani, D. D. S. R. , Rui, B. R. , Kawai, G. K. V. , Losano, J. D. A. , & Vannucchi, C. I. (2020). Effect of senescence on morphological, functional and oxidative features of fresh and cryopreserved canine sperm. Aging Male, 23(4), 279–286. 10.1080/13685538.2018.1487931 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

