Lactate-Loaded Nanoparticles Induce Glioma Cytotoxicity and Increase the Survival of Rats Bearing Malignant Glioma Brain Tumor
- PMID: 35214059
- PMCID: PMC8880216
- DOI: 10.3390/pharmaceutics14020327
Lactate-Loaded Nanoparticles Induce Glioma Cytotoxicity and Increase the Survival of Rats Bearing Malignant Glioma Brain Tumor
Abstract
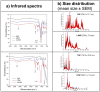
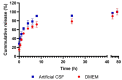
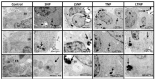
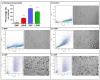
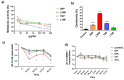
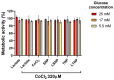
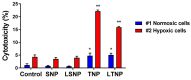
A glioblastoma is an aggressive form of a malignant glial-derived tumor with a poor prognosis despite multimodal therapy approaches. Lactate has a preponderant role in the tumor microenvironment, playing an immunoregulatory role as well as being a carbon source for tumor growth. Lactate homeostasis depends on the proper functioning of intracellular lactate regulation systems, such as transporters and enzymes involved in its synthesis and degradation, with evidence that an intracellular lactate overload generates metabolic stress on tumor cells and tumor cell death. We propose that the delivery of a lactate overload carried in nanoparticles, allowing the intracellular release of lactate, would compromise the survival of tumor cells. We synthesized and characterized silica and titania nanoparticles loaded with lactate to evaluate the cellular uptake, metabolic activity, pH modification, and cytotoxicity on C6 cells under normoxia and chemical hypoxia, and, finally, determined the survival of an orthotopic malignant glioma model after in situ administration. A dose-dependent reduction in metabolic activity of treated cells under normoxia was found, but not under hypoxia, independent of glucose concentration. Lactated-loaded silica nanoparticles were highly cytotoxic (58.1% of dead cells) and generated significant supernatant acidification. In vivo, lactate-loaded silica nanoparticles significantly increased the median survival time of malignant glioma-bearing rats (p = 0.005) when administered in situ. These findings indicate that lactate-loaded silica nanoparticles are cytotoxic on glioma cells in vitro and in vivo.
Keywords: cytotoxicity; glioma; in situ therapy; lactate-loaded nanoparticle; metabolism.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Convection enhanced delivery of cisplatin-loaded brain penetrating nanoparticles cures malignant glioma in rats.J Control Release. 2017 Oct 10;263:112-119. doi: 10.1016/j.jconrel.2017.03.007. Epub 2017 Mar 7. J Control Release. 2017. PMID: 28279797 Free PMC article.
-
Targeted delivery of vitamin D3-loaded nanoparticles to C6 glioma cell line increased resistance to doxorubicin, epirubicin, and docetaxel in vitro.In Vitro Cell Dev Biol Anim. 2016 Dec;52(10):989-1000. doi: 10.1007/s11626-016-0072-7. Epub 2016 Aug 8. In Vitro Cell Dev Biol Anim. 2016. PMID: 27503515
-
LDLR-mediated peptide-22-conjugated nanoparticles for dual-targeting therapy of brain glioma.Biomaterials. 2013 Dec;34(36):9171-82. doi: 10.1016/j.biomaterials.2013.08.039. Epub 2013 Sep 3. Biomaterials. 2013. PMID: 24008043
-
The Acidic Brain-Glycolytic Switch in the Microenvironment of Malignant Glioma.Int J Mol Sci. 2021 May 24;22(11):5518. doi: 10.3390/ijms22115518. Int J Mol Sci. 2021. PMID: 34073734 Free PMC article. Review.
-
Drug-loaded nanoparticle systems and adult stem cells: a potential marriage for the treatment of malignant glioma?Oncotarget. 2013 Mar;4(3):378-96. doi: 10.18632/oncotarget.937. Oncotarget. 2013. PMID: 23594406 Free PMC article. Review.
Cited by
-
Current Non-Metal Nanoparticle-Based Therapeutic Approaches for Glioblastoma Treatment.Biomedicines. 2024 Aug 11;12(8):1822. doi: 10.3390/biomedicines12081822. Biomedicines. 2024. PMID: 39200286 Free PMC article. Review.
-
How Warburg-Associated Lactic Acidosis Rewires Cancer Cell Energy Metabolism to Resist Glucose Deprivation.Cancers (Basel). 2023 Feb 23;15(5):1417. doi: 10.3390/cancers15051417. Cancers (Basel). 2023. PMID: 36900208 Free PMC article. Review.
-
CircXPO1 Promotes Glioblastoma Malignancy by Sponging miR-7-5p.Cells. 2023 Mar 8;12(6):831. doi: 10.3390/cells12060831. Cells. 2023. PMID: 36980172 Free PMC article.
-
Engineering Poly(lactic Acid)-Based Scaffolds for Abundant, Sustained, and Prolonged Lactate Release.ACS Polym Au. 2025 Mar 25;5(3):247-260. doi: 10.1021/acspolymersau.4c00097. eCollection 2025 Jun 11. ACS Polym Au. 2025. PMID: 40519951 Free PMC article.
-
Melatonin in Combination with Albendazole or Albendazole Sulfoxide Produces a Synergistic Cytotoxicity against Malignant Glioma Cells through Autophagy and Apoptosis.Brain Sci. 2023 May 27;13(6):869. doi: 10.3390/brainsci13060869. Brain Sci. 2023. PMID: 37371349 Free PMC article.
References
-
- Fernandes C., Costa A., Osório L., Lago R.C., Linhares P., Carvalho B., Caeiro C. Current Standards of Care in Glioblastoma Therapy. In: De Vleeschouwer S., editor. Glioblastoma. Codon Publications; Brisbane, Australia: 2017. pp. 197–241. - PubMed
-
- Guyon J., Romero-Garmendia I., Bikfalvi A., Daubon T. FSMP-14. Metabolic symbiosis in glioblastoma: Lactate dehydrogenases take the lead. Neuro Oncol. Adv. 2021;3:i19. doi: 10.1093/noajnl/vdab024.078. - DOI
-
- Nikoobakht M., Shamshiripour P., Azimi Nekoo Z., Fallah Haghmohammadi S. Elevated Lactate and Total Protein Levels in Stereotactic Brain Biopsy Specimen; Potential Biomarkers of Malignancy and Poor Prognosis. Arch. Iran. Med. 2019;22:125–131. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

