Fabrication and Integration of Functionalized N-rGO-Ni/Ag and N-rGO-Ni/Co Nanocomposites as Synergistic Oxygen Electrocatalysts in Fuel Cells
- PMID: 35214913
- PMCID: PMC8877386
- DOI: 10.3390/nano12040585
Fabrication and Integration of Functionalized N-rGO-Ni/Ag and N-rGO-Ni/Co Nanocomposites as Synergistic Oxygen Electrocatalysts in Fuel Cells
Abstract
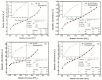
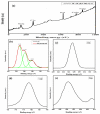
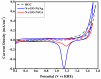
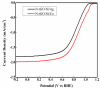
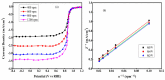
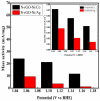
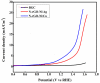
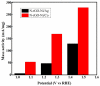
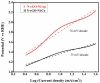
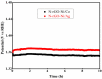
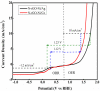
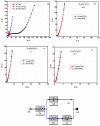
Fabrication of composites by developing simple techniques can be an efficient way to modify the desire properties of the materials. This paper presents a detailed study on synthesis of low cost and efficient nitrogen doped reduced graphene oxide nickle-silver (N-rGO-Ni/Ag) and nickel-cobalt (N-rGO-Ni/Co) nanocomposites as electrocatalysts in fuel cell using one-pot blended reflux condensation route. An admirable correlation in the structures and properties of the synthesized nanocomposites was observed. The Oxygen Reduction Reaction (ORR) values for N-rGO-Ni/Ag and N-rGO-Ni/Co calculated from the onset potential, using Linear Sweep Voltammetry (LSV), were found to be 1.096 and 1.146. While the half wave potential were determined to be 1.046 and 1.106, respectively, N-rGO-Ni/Ag and N-rGO-Ni/Co. The Tafel and bi-functional (ORR/OER) values were calculated as 76 and 35 mV/decade and 1.23 and 1.12 V, respectively, for N-rGO-Ni/Ag and N-rGO-Ni/Co. The lower onset and half wave potential, low charge transfer resistance (Rct = 1.20 Ω/cm2) and internal solution resistance (Rs = 8.84 × 10-1 Ω/cm2), lower Tafel values (35 mV), satisfactory LSV measurements and mass activity (24.5 at 1.056 V for ORR and 54.9 at 1.056 for OER) demonstrate the remarkable electrocatalytic activity of N-rGO-Ni/Co for both ORR and OER. The chronamperometric stability for synthesized nanocomposites was found satisfactory up to 10 h.
Keywords: N-doped reduced graphene oxide; bi-functional electrocatalysis; oxygen evolution reaction; oxygen reduction reaction.
Conflict of interest statement
Authors declare no conflict of interest.
Figures



Similar articles
-
Co3O4 nanoparticles anchored on nitrogen-doped reduced graphene oxide as a multifunctional catalyst for H2O2 reduction, oxygen reduction and evolution reaction.Sci Rep. 2017 Mar 8;7:43638. doi: 10.1038/srep43638. Sci Rep. 2017. PMID: 28272415 Free PMC article.
-
CuCo2S4@B,N-Doped Reduced Graphene Oxide Hybrid as a Bifunctional Electrocatalyst for Oxygen Reduction and Evolution Reactions.ACS Omega. 2022 May 31;7(23):19183-19192. doi: 10.1021/acsomega.2c00183. eCollection 2022 Jun 14. ACS Omega. 2022. PMID: 35721942 Free PMC article.
-
Novel Two-Dimensional Carbon-Chromium Nitride-Based Composite as an Electrocatalyst for Oxygen Reduction Reaction.Front Chem. 2019 Nov 12;7:738. doi: 10.3389/fchem.2019.00738. eCollection 2019. Front Chem. 2019. PMID: 31781536 Free PMC article.
-
Rod-like nickel doped Co3Se4/reduced graphene oxide hybrids as efficient electrocatalysts for oxygen evolution reactions.Nanoscale. 2021 Feb 14;13(6):3698-3708. doi: 10.1039/d0nr08591j. Epub 2021 Feb 5. Nanoscale. 2021. PMID: 33543742
-
Facile Synthesis of Low-Cost Copper-Silver and Cobalt-Silver Alloy Nanoparticles on Reduced Graphene Oxide as Efficient Electrocatalysts for Oxygen Reduction Reaction in Alkaline Media.Nanomaterials (Basel). 2022 Aug 2;12(15):2657. doi: 10.3390/nano12152657. Nanomaterials (Basel). 2022. PMID: 35957088 Free PMC article.
Cited by
-
Facile Fabrication of Nickel Supported on Reduced Graphene Oxide Composite for Oxygen Reduction Reaction.Nanomaterials (Basel). 2023 Dec 5;13(24):3087. doi: 10.3390/nano13243087. Nanomaterials (Basel). 2023. PMID: 38132985 Free PMC article.
-
Editorial for the Special Issue on "Graphene-Related Materials: Synthesis and Applications".Nanomaterials (Basel). 2022 Aug 10;12(16):2740. doi: 10.3390/nano12162740. Nanomaterials (Basel). 2022. PMID: 36014605 Free PMC article.
References
-
- Farooqui U., Ahmad A., Hamid N. Challenges and potential advantages of membranes in lithium air batteries: A review. Renew. Sustain. Energy Rev. 2017;77:1114–1129. doi: 10.1016/j.rser.2016.11.220. - DOI
-
- Othman R., Dicks A.L., Zhu Z. Non precious metal catalysts for the PEM fuel cell cathode. Int. J. Hydrogen Energy. 2012;37:357–372. doi: 10.1016/j.ijhydene.2011.08.095. - DOI
-
- Zhao H., Wang J., Chen C., Chen D., Gao Y., Saccoccio M., Ciucci F. A bi-functional catalyst for oxygen reduction and oxygen evolution reactions from used baby diapers: α-Fe2O3 wrapped in P and S dual doped graphitic carbon. RSC Adv. 2016;6:64258–64265. doi: 10.1039/C6RA11352D. - DOI
LinkOut - more resources
Full Text Sources

