Oligosaccharides Ameliorate Acute Kidney Injury by Alleviating Cluster of Differentiation 44-Mediated Immune Responses in Renal Tubular Cells
- PMID: 35215410
- PMCID: PMC8877265
- DOI: 10.3390/nu14040760
Oligosaccharides Ameliorate Acute Kidney Injury by Alleviating Cluster of Differentiation 44-Mediated Immune Responses in Renal Tubular Cells
Abstract
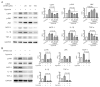
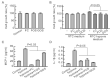
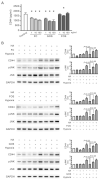
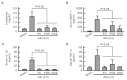
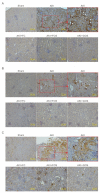
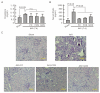
Acute kidney injury (AKI) is a sudden episode of kidney damage that commonly occurs in patients admitted to hospitals. To date, no ideal treatment has been developed to reduce AKI severity. Oligo-fucoidan (FC) interferes with renal tubular cell surface protein cluster of differentiation 44 (CD44) to prevent renal interstitial fibrosis; however, the influence of oligosaccharides on AKI remains unknown. In this study, FC, galacto-oligosaccharide (GOS), and fructo-oligosaccharide (FOS) were selected to investigate the influence of oligosaccharides on AKI. All three oligosaccharides have been proven to be partially absorbed by the intestine. We found that the oligosaccharides dose-dependently reduced CD44 antigenicity and suppressed the hypoxia-induced expression of CD44, phospho-JNK, MCP-1, IL-1β, and TNF-α in NRK-52E renal tubular cells. Meanwhile, CD44 siRNA transfection and JNK inhibitor SP600125 reduced the hypoxia-induced expression of phospho-JNK and cytokines. The ligand of CD44, hyaluronan, counteracted the influence of oligosaccharides on CD44 and phospho-JNK. At 2 days post-surgery for ischemia-reperfusion injury, oligosaccharides reduced kidney inflammation, serum creatine, MCP-1, IL-1β, and TNF-α in AKI mice. At 7 days post-surgery, kidney recovery was promoted. These results indicate that FC, GOS, and FOS inhibit the hypoxia-induced CD44/JNK cascade and cytokines in renal tubular cells, thereby ameliorating AKI and kidney inflammation in AKI mice. Therefore, oligosaccharide supplementation is a potential healthcare strategy for patients with AKI.
Keywords: acute kidney injury; cluster of differentiation 44; fructo-oligosaccharide; galacto-oligosaccharide; inflammation; oligo-fucoidan.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Joannidis M., Druml W., Forni L.G., Groeneveld A.B.J., Honore P.M., Hoste E., Ostermann M., Oudemans-van Straaten H.M., Schetz M. Prevention of acute kidney injury and protection of renal function in the intensive care unit: Update 2017: Expert opinion of the Working Group on Prevention, AKI section, European Society of Intensive Care Medicine. Intensive Care Med. 2017;43:730–749. doi: 10.1007/s00134-017-4832-y. - DOI - PMC - PubMed
-
- Bijuklic K., Jennings P., Kountchev J., Hasslacher J., Aydin S., Sturn D., Pfaller W., Patsch J.R., Joannidis M. Migration of leukocytes across an endothelium-epithelium bilayer as a model of renal interstitial inflammation. Am. J. Physiol. Physiol. 2007;293:C486–C492. doi: 10.1152/ajpcell.00419.2006. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

