Elucidation of the Mechanism of Host NMD Suppression by HTLV-1 Rex: Dissection of Rex to Identify the NMD Inhibitory Domain
- PMID: 35215946
- PMCID: PMC8875924
- DOI: 10.3390/v14020344
Elucidation of the Mechanism of Host NMD Suppression by HTLV-1 Rex: Dissection of Rex to Identify the NMD Inhibitory Domain
Abstract
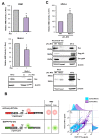
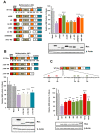
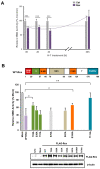
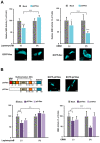
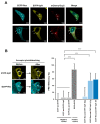
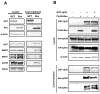
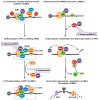
The human retrovirus human T-cell leukemia virus type I (HTLV-1) infects human T cells by vertical transmission from mother to child through breast milk or horizontal transmission through blood transfusion or sexual contact. Approximately 5% of infected individuals develop adult T-cell leukemia/lymphoma (ATL) with a poor prognosis, while 95% of infected individuals remain asymptomatic for the rest of their lives, during which time the infected cells maintain a stable immortalized latent state in the body. It is not known why such a long latent state is maintained. We hypothesize that the role of functional proteins of HTLV-1 during early infection influences the phenotype of infected cells in latency. In eukaryotic cells, a mRNA quality control mechanism called nonsense-mediated mRNA decay (NMD) functions not only to eliminate abnormal mRNAs with nonsense codons but also to target virus-derived RNAs. We have reported that HTLV-1 genomic RNA is a potential target of NMD, and that Rex suppresses NMD and stabilizes viral RNA against it. In this study, we aimed to elucidate the molecular mechanism of NMD suppression by Rex using various Rex mutant proteins. We found that region X (aa20-57) of Rex, the function of which has not been clarified, is required for NMD repression. We showed that Rex binds to Upf1, which is the host key regulator to detect abnormal mRNA and initiate NMD, through this region. Rex also interacts with SMG5 and SMG7, which play essential roles for the completion of the NMD pathway. Moreover, Rex selectively binds to Upf3B, which is involved in the normal NMD complex, and replaces it with a less active form, Upf3A, to reduce NMD activity. These results revealed that Rex invades the NMD cascade from its initiation to completion and suppresses host NMD activity to protect the viral genomic mRNA.
Keywords: HTLV-1 rex; NMD inhibition; SMG5/7; Upf1; Upf3; viral RNA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Poiesz B.J., Ruscetti F.W., Gazdar A.F., Bunn P.A., Minna J.D., Gallo R.C. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc. Natl. Acad. Sci. USA. 1980;77:7415–7419. doi: 10.1073/pnas.77.12.7415. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

