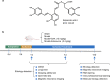
Long-term administration of salvianolic acid A promotes endogenous neurogenesis in ischemic stroke rats through activating Wnt3a/GSK3β/β-catenin signaling pathway
- PMID: 35217812
- PMCID: PMC9433393
- DOI: 10.1038/s41401-021-00844-9
Long-term administration of salvianolic acid A promotes endogenous neurogenesis in ischemic stroke rats through activating Wnt3a/GSK3β/β-catenin signaling pathway
Abstract
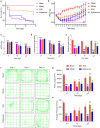
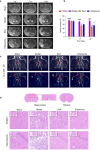
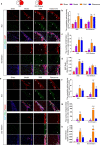
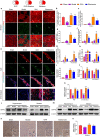
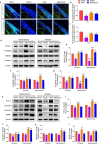
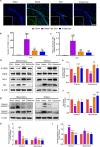
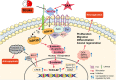
Stroke is the major cause of death and disability worldwide. Most stroke patients who survive in the acute phase of ischemia display various extents of neurological deficits. In order to improve the prognosis of ischemic stroke, promoting endogenous neurogenesis has attracted great attention. Salvianolic acid A (SAA) has shown neuroprotective effects against ischemic diseases. In the present study, we investigated the neurogenesis effects of SAA in ischemic stroke rats, and explored the underlying mechanisms. An autologous thrombus stroke model was established by electrocoagulation. The rats were administered SAA (10 mg/kg, ig) or a positive drug edaravone (5 mg/kg, iv) once a day for 14 days. We showed that SAA administration significantly decreased infarction volume and vascular embolism, and ameliorated pathological injury in the hippocampus and striatum as well as the neurological deficits as compared with the model rats. Furthermore, we found that SAA administration significantly promoted neural stem/progenitor cells (NSPCs) proliferation, migration and differentiation into neurons, enhanced axonal regeneration and diminished neuronal apoptosis around the ipsilateral subventricular zone (SVZ), resulting in restored neural density and reconstructed neural circuits in the ischemic striatum. Moreover, we revealed that SAA-induced neurogenesis was associated to activating Wnt3a/GSK3β/β-catenin signaling pathway and downstream target genes in the hippocampus and striatum. Edaravone exerted equivalent inhibition on neuronal apoptosis in the SVZ, as SAA, but edaravone-induced neurogenesis was weaker than that of SAA. Taken together, our results demonstrate that long-term administration of SAA improves neurological function through enhancing endogenous neurogenesis and inhibiting neuronal apoptosis in ischemic stroke rats via activating Wnt3a/GSK3β/β-catenin signaling pathway. SAA may be a potential therapeutic drug to promote neurogenesis after stroke.
Keywords: Wnt3a/GSK3β/β-catenin signaling; hippocampus; ischemic stroke; neurogenesis; salvianolic acid A; striatum.
© 2022. The Author(s), under exclusive licence to CPS and SIMM.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Salvianolic acid A alleviates ischemic brain injury through the inhibition of inflammation and apoptosis and the promotion of neurogenesis in mice.Free Radic Biol Med. 2016 Oct;99:508-519. doi: 10.1016/j.freeradbiomed.2016.09.006. Epub 2016 Sep 5. Free Radic Biol Med. 2016. PMID: 27609227
-
Salvianolic acid A prevented cerebrovascular endothelial injury caused by acute ischemic stroke through inhibiting the Src signaling pathway.Acta Pharmacol Sin. 2021 Mar;42(3):370-381. doi: 10.1038/s41401-020-00568-2. Epub 2020 Dec 10. Acta Pharmacol Sin. 2021. PMID: 33303991 Free PMC article.
-
XQ-1H alleviates cerebral ischemia in mice through inhibition of apoptosis and promotion of neurogenesis in a Wnt/β-catenin signaling dependent way.Life Sci. 2019 Oct 15;235:116844. doi: 10.1016/j.lfs.2019.116844. Epub 2019 Sep 6. Life Sci. 2019. PMID: 31499069
-
Unravelling neuroregenerative and neuroprotective roles of Wnt/β-catenin pathway in ischemic stroke: Insights into molecular mechanisms.Neuroscience. 2025 Jan 26;565:527-547. doi: 10.1016/j.neuroscience.2024.12.024. Epub 2024 Dec 15. Neuroscience. 2025. PMID: 39681254 Review.
-
Therapeutic Strategies for Ischemic Stroke: Modulating the Adult Neural Stem Cell Niche through the Wnt/β-catenin Pathway.J Integr Neurosci. 2024 Jul 15;23(7):131. doi: 10.31083/j.jin2307131. J Integr Neurosci. 2024. PMID: 39082287 Review.
Cited by
-
Bioactive compounds from Chinese herbal plants for neurological health: mechanisms, pathways, and functional food applications.Front Nutr. 2025 Jan 31;12:1537363. doi: 10.3389/fnut.2025.1537363. eCollection 2025. Front Nutr. 2025. PMID: 39957765 Free PMC article. Review.
-
Alpha‑Asarone Ameliorates Neuronal Injury After Ischemic Stroke and Hemorrhagic Transformation by Attenuating Blood-Brain Barrier Destruction, Promoting Neurogenesis, and Inhibiting Neuroinflammation.Mol Neurobiol. 2025 Apr;62(4):5252-5272. doi: 10.1007/s12035-024-04596-5. Epub 2024 Nov 12. Mol Neurobiol. 2025. PMID: 39531192
-
Meldonium, as a potential neuroprotective agent, promotes neuronal survival by protecting mitochondria in cerebral ischemia-reperfusion injury.J Transl Med. 2024 Aug 15;22(1):771. doi: 10.1186/s12967-024-05222-7. J Transl Med. 2024. PMID: 39148053 Free PMC article.
-
Nanocarriers Loaded with Danshensu for Treating Ischemic Stroke by Reducing Oxidative Stress and Glial Overactivation.ACS Omega. 2024 Aug 5;9(33):35686-35694. doi: 10.1021/acsomega.4c03991. eCollection 2024 Aug 20. ACS Omega. 2024. PMID: 39184494 Free PMC article.
-
Mitochondrion-based organellar therapies for central nervous system diseases.Cell Commun Signal. 2024 Oct 10;22(1):487. doi: 10.1186/s12964-024-01843-z. Cell Commun Signal. 2024. PMID: 39390521 Free PMC article. Review.
References
-
- Yamashita T, Ninomiya M, Hernández Acosta P, García-Verdugo JM, Sunabori T, Sakaguchi M, et al. Subventricular zone-derived neuroblasts migrate and differentiate into mature neurons in the post-stroke adult striatum. J Neurosci. 2006;26:6627–36. doi: 10.1523/JNEUROSCI.0149-06.2006. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

