Loss of Neuron Navigator 2 Impairs Brain and Cerebellar Development
- PMID: 35218524
- PMCID: PMC9985553
- DOI: 10.1007/s12311-022-01379-3
Loss of Neuron Navigator 2 Impairs Brain and Cerebellar Development
Abstract
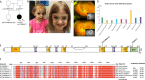
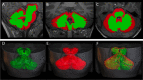
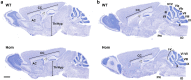
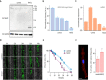
Cerebellar hypoplasia and dysplasia encompass a group of clinically and genetically heterogeneous disorders frequently associated with neurodevelopmental impairment. The Neuron Navigator 2 (NAV2) gene (MIM: 607,026) encodes a member of the Neuron Navigator protein family, widely expressed within the central nervous system (CNS), and particularly abundant in the developing cerebellum. Evidence across different species supports a pivotal function of NAV2 in cytoskeletal dynamics and neurite outgrowth. Specifically, deficiency of Nav2 in mice leads to cerebellar hypoplasia with abnormal foliation due to impaired axonal outgrowth. However, little is known about the involvement of the NAV2 gene in human disease phenotypes. In this study, we identified a female affected with neurodevelopmental impairment and a complex brain and cardiac malformations in which clinical exome sequencing led to the identification of NAV2 biallelic truncating variants. Through protein expression analysis and cell migration assay in patient-derived fibroblasts, we provide evidence linking NAV2 deficiency to cellular migration deficits. In model organisms, the overall CNS histopathology of the Nav2 hypomorphic mouse revealed developmental anomalies including cerebellar hypoplasia and dysplasia, corpus callosum hypo-dysgenesis, and agenesis of the olfactory bulbs. Lastly, we show that the NAV2 ortholog in Drosophila, sickie (sick) is widely expressed in the fly brain, and sick mutants are mostly lethal with surviving escapers showing neurobehavioral phenotypes. In summary, our results unveil a novel human neurodevelopmental disorder due to genetic loss of NAV2, highlighting a critical conserved role of the NAV2 gene in brain and cerebellar development across species.
Keywords: Axon elongation, Brain malformation; Cerebellar cortical dysplasia; Cerebellar hypoplasia; NAV2; Neuron migration.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Accogli A, Addour-Boudrahem N, Srour M. Diagnostic Approach to Cerebellar Hypoplasia. Cerebellum. 2021;20(4):631–58. 10.1007/s12311-020-01224-5. - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

