Cordyceps cicadae Ameliorates Renal Hypertensive Injury and Fibrosis Through the Regulation of SIRT1-Mediated Autophagy
- PMID: 35222012
- PMCID: PMC8866973
- DOI: 10.3389/fphar.2021.801094
Cordyceps cicadae Ameliorates Renal Hypertensive Injury and Fibrosis Through the Regulation of SIRT1-Mediated Autophagy
Abstract
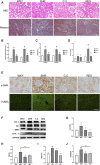
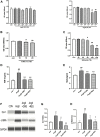
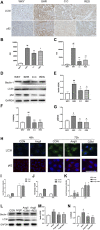
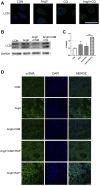
Hypertensive renal injury is a complication of hypertension. Cordyceps cicadae (C. cicadae) is a traditional Chinese medicine used to treat chronic kidney diseases especially renal fibrosis. Autophagy is described as a cell self-renewal process that requires lysosomal degradation and is utilized for the maintenance of cellular energy homeostasis. The present study explores the mechanism underlying C. cicadae's renoprotection on hypertensive nephropathy (HN). First, HN rat models were established on spontaneously hypertensive rats (SHRs). The expression of fibrosis-related protein and autophagy-associated protein was detected in vivo. NRK-52E cells exposed to AngII were chosen to observe the potential health benefits of C. cicadae on renal damage. The level of extracellular matrix accumulation was detected using capillary electrophoresis immunoquantification and immunohistochemistry. After treatment with lysosomal inhibitors (chloroquine) or an autophagy activator (rapamycin), the expression of Beclin-1, LC3II, and SQSTM1/p62 was further investigated. The study also investigated the change in sirtuin1 (SIRT1), fork head box O3a (FOXO3a), and peroxidation (superoxide dismutase (SOD) and malondialdehyde (MDA)) expression when intervened by resveratrol. The changes in SIRT1 and FOXO3a were measured in patients and the SHRs. Here, we observed that C. cicadae significantly decreased damage to renal tubular epithelial cells and TGFβ1, α-smooth muscle actin (α-SMA), collagen I (Col-1), and fibronectin expression. Meanwhile, autophagy defects were observed both in vivo and in vitro. C. cicadae intervention significantly downregulated Beclin-1 and LC3II and decreased SQSTM1/p62, showing an inhibition of autophagic vesicles and the alleviation of autophagy stress. These functions were suppressed by rapamycin, and the results were just as effective as the resveratrol treatment. HN patients and the SHRs exhibited decreased levels of SIRT1 and FOXO3a. We also observed a positive correlation between SIRT1/FOXO3a and antifibrotic effects. Similar to the resveratrol group, the expression of SIRT1/FOXO3a and oxidative stress were elevated by C. cicadae in vivo. Taken together, our findings show that C. cicadae ameliorates tubulointerstitial fibrosis and delays HN progression. Renoprotection was likely attributable to the regulation of autophagic stress mediated by the SIRT1 pathway and achieved by regulating FOXO3a and oxidative stress.
Keywords: Cordyceps cicadae; SIRT1; autophagy; fibrosis; hypertensive renal injury.
Copyright © 2022 Cai, Feng, Jia, Guo, Zhang, Zhao, Wang, Liu and Liu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Cordyceps cicadae Prevents Renal Tubular Epithelial Cell Apoptosis by Regulating the SIRT1/p53 Pathway in Hypertensive Renal Injury.Evid Based Complement Alternat Med. 2020 Jan 13;2020:7202519. doi: 10.1155/2020/7202519. eCollection 2020. Evid Based Complement Alternat Med. 2020. PMID: 32419819 Free PMC article.
-
Cordyceps cicadae ameliorates inflammatory responses, oxidative stress, and fibrosis by targeting the PI3K/mTOR-mediated autophagy pathway in the renal of MRL/lpr mice.Immun Inflamm Dis. 2024 Jan;12(1):e1168. doi: 10.1002/iid3.1168. Immun Inflamm Dis. 2024. PMID: 38270299 Free PMC article.
-
Clerodendranthus spicatus-Cordyceps cicadae regulates mitophagy and protects renal tubular epithelial cells from hyperuricemic nephropathy.J Ethnopharmacol. 2025 Jun 12;349:119926. doi: 10.1016/j.jep.2025.119926. Epub 2025 May 5. J Ethnopharmacol. 2025. PMID: 40334761
-
Can Cordyceps cicadae be used as an alternative to Cordyceps militaris and Cordyceps sinensis? - A review.J Ethnopharmacol. 2020 Jul 15;257:112879. doi: 10.1016/j.jep.2020.112879. Epub 2020 Apr 16. J Ethnopharmacol. 2020. PMID: 32305637 Review.
-
Potential Therapeutic Strategies for Renal Fibrosis: Cordyceps and Related Products.Front Pharmacol. 2022 Jul 8;13:932172. doi: 10.3389/fphar.2022.932172. eCollection 2022. Front Pharmacol. 2022. PMID: 35873549 Free PMC article. Review.
Cited by
-
Use of bailing capsules (cordyceps sinensis) in the treatment of chronic kidney disease: a meta-analysis and network pharmacology.Front Pharmacol. 2024 Apr 5;15:1342831. doi: 10.3389/fphar.2024.1342831. eCollection 2024. Front Pharmacol. 2024. PMID: 38645562 Free PMC article. Review.
-
LYC inhibits the AKT signaling pathway to activate autophagy and ameliorate TGFB-induced renal fibrosis.Autophagy. 2024 May;20(5):1114-1133. doi: 10.1080/15548627.2023.2287930. Epub 2023 Nov 30. Autophagy. 2024. PMID: 38037248 Free PMC article.
-
JiangyaTongluo decoction ameliorates tubulointerstitial fibrosis via regulating the SIRT1/PGC-1α/mitophagy axis in hypertensive nephropathy.Front Pharmacol. 2024 Dec 12;15:1491315. doi: 10.3389/fphar.2024.1491315. eCollection 2024. Front Pharmacol. 2024. PMID: 39726785 Free PMC article.
-
A novel link between silent information regulator 1 and autophagy in cerebral ischemia-reperfusion.Front Neurosci. 2022 Nov 23;16:1040182. doi: 10.3389/fnins.2022.1040182. eCollection 2022. Front Neurosci. 2022. PMID: 36507335 Free PMC article. Review.
-
Advance on Chinese Medicine for Hypertensive Renal Damage: Focus on the Complex Molecular Mechanisms.Chin J Integr Med. 2024 Oct;30(10):938-948. doi: 10.1007/s11655-024-3662-3. Epub 2024 Jul 3. Chin J Integr Med. 2024. PMID: 38958884 Review.
References
-
- Aibara Y., Nakashima A., Kawano K. I., Yusoff F. M., Mizuki F., Kishimoto S., et al. (2020). Daily Low-Intensity Pulsed Ultrasound Ameliorates Renal Fibrosis and Inflammation in Experimental Hypertensive and Diabetic Nephropathy. Hypertension 76 (6), 1906–1914. 10.1161/HYPERTENSIONAHA.120.15237 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

