Methodology for Cross-Talk Elimination in Simultaneous Voltage and Calcium Optical Mapping Measurements With Semasbestic Wavelengths
- PMID: 35222080
- PMCID: PMC8874316
- DOI: 10.3389/fphys.2022.812968
Methodology for Cross-Talk Elimination in Simultaneous Voltage and Calcium Optical Mapping Measurements With Semasbestic Wavelengths
Abstract
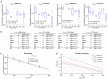
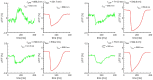
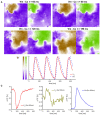
Most cardiac arrhythmias at the whole heart level result from alteration of cell membrane ionic channels and intracellular calcium concentration ([Ca2+] i ) cycling with emerging spatiotemporal behavior through tissue-level coupling. For example, dynamically induced spatial dispersion of action potential duration, QT prolongation, and alternans are clinical markers for arrhythmia susceptibility in regular and heart-failure patients that originate due to changes of the transmembrane voltage (V m) and [Ca2+] i . We present an optical-mapping methodology that permits simultaneous measurements of the V m - [Ca2+] i signals using a single-camera without cross-talk, allowing quantitative characterization of favorable/adverse cell and tissue dynamical effects occurring from remodeling and/or drugs in heart failure. We demonstrate theoretically and experimentally in six different species the existence of a family of excitation wavelengths, we termed semasbestic, that give no change in signal for one dye, and thus can be used to record signals from another dye, guaranteeing zero cross-talk.
Keywords: alternans; fluorescent dyes; intracellular free calcium concentration; isosbestic point; optical mapping; semasbestic wavelength; transmembrane voltage.
Copyright © 2022 Uzelac, Crowley, Iravanian, Kim, Cho and Fenton.
Conflict of interest statement
IU is the owner of Aleksa Tech Inc., a manufacturer of power sources for LED illumination in optical mapping measurements. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Bachtel A. D., Gray R. A., Stohlman J. M., Bourgeois E. B., Pollard A. E., Rogers J. M. (2011). A novel approach to dual excitation ratiometric optical mapping of cardiac action potentials with di-4-anepps using pulsed led excitation. IEEE Trans. Biomed. Eng. 58, 2120–2126. 10.1109/TBME.2011.2148719 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

