Sleep Disorders in Patients With Craniopharyngioma: A Physiopathological and Practical Update
- PMID: 35222233
- PMCID: PMC8863754
- DOI: 10.3389/fneur.2021.817257
Sleep Disorders in Patients With Craniopharyngioma: A Physiopathological and Practical Update
Abstract
Sleep disorders (SDs) represent an important issue in patients with craniopharyngioma (CP). Nearly 70% of these patients complain of sleep-wake cycle alterations and/or excessive diurnal somnolence due to sleep-related breathing disorders, such as obstructive sleep apnea (OSA) and/or central hypersomnia, including secondary narcolepsy. SDs may severely reduce quality of life, increase disease-related cardiorespiratory and cardiovascular morbidity, and finally play a major role in increased long-term mortality reported on patients with CP. A major risk factor for SDs is represented by the hypothalamic syndrome, which may develop because of direct hypothalamic damage by the tumor itself and/or complications of the treatments, neurosurgery and/or radiotherapy, and typically includes permanent neuroendocrine dysfunctions, morbid obesity, and secondary metabolic disorders. Despite increasing attention to SDs in the general population, and in particular to OSA as a risk factor for cardio-metabolic diseases and excessive daytime somnolence, sleep evaluation is still not routinely proposed to patients with CP. Hence, SDs are often underdiagnosed and undertreated. The aim of this paper is to update current knowledge of the pathogenesis and prevalence of SDs in patients with CP and propose practical algorithms for their evaluation and management in clinical practice. Particular attention is paid to screening and diagnostic tools for appropriate characterization of SDs, identification of risk factors, and potential role of hypothalamic sparing surgery in the prevention of morbid obesity and SDs. Available tools in sleep medicine, including lifestyle interventions, drugs, and respiratory devices, are discussed, as well as the importance of optimal hormone replacement and metabolic interventions. Current limits in the diagnosis and treatment of SDs in patients with CP and possible future avenues for research agenda are also considered.
Keywords: circadian rythm disorders; craniopharyngioma; hypersomnia; hypothalamic obesity; hypothalamic syndrome; narcolepsy; obstructive sleep apnea; sleep disorder.
Copyright © 2022 Romigi, Feola, Cappellano, De Angelis, Pio, Caccamo, Testa, Vitrani, Centonze, Colonnese, Esposito and Jaffrain-Rea.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

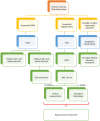
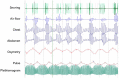
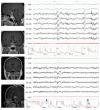
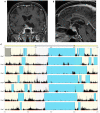
References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

