Establishment of Chimerism and Organ Transplant Tolerance in Laboratory Animals: Safety and Efficacy of Adaptation to Humans
- PMID: 35222384
- PMCID: PMC8866443
- DOI: 10.3389/fimmu.2022.805177
Establishment of Chimerism and Organ Transplant Tolerance in Laboratory Animals: Safety and Efficacy of Adaptation to Humans
Abstract
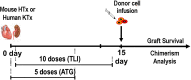
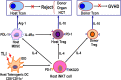
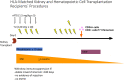
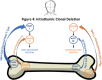
The definition of immune tolerance to allogeneic tissue and organ transplants in laboratory animals and humans continues to be the acceptance of the donor graft, rejection of third-party grafts, and specific unresponsiveness of recipient immune cells to the donor alloantigens in the absence of immunosuppressive treatments. Actively acquired tolerance was achieved in mice more than 60 years ago by the establishment of mixed chimerism in neonatal mice. Once established, mixed chimerism was self-perpetuating and allowed for acceptance of tissue transplants in adults. Successful establishment of tolerance in humans has now been reported in several clinical trials based on the development of chimerism after combined transplantation of hematopoietic cells and an organ from the same donor. This review examines the mechanisms of organ graft acceptance after establishment of mixed chimerism (allo-tolerance) or complete chimerism (self-tolerance), and compares the development of graft versus host disease (GVHD) and graft versus tumor (GVT) activity in complete and mixed chimerism. GVHD, GVT activity, and complete chimerism are also discussed in the context of bone marrow transplantation to treat hematologic malignancies. The roles of transient versus persistent mixed chimerism in the induction and maintenance of tolerance and organ graft acceptance in animal models and clinical studies are compared. Key differences in the stability of mixed chimeras and tolerance induction in MHC matched and mismatched rodents, large laboratory animals, and humans are examined to provide insights into the safety and efficacy of translation of results of animal models to clinical trials.
Keywords: animal models; chimerism; human trials; immune suppression; transplant tolerance.
Copyright © 2022 Lowsky and Strober.
Conflict of interest statement
RL and SS are scientific founders of Medeor Therapeutics. SS is a consult to Medeor Therapeutics.
Figures
Similar articles
-
The importance of MHC class II in allogeneic bone marrow transplantation and chimerism-based solid organ tolerance in a rat model.PLoS One. 2020 May 22;15(5):e0233497. doi: 10.1371/journal.pone.0233497. eCollection 2020. PLoS One. 2020. PMID: 32442182 Free PMC article.
-
Facilitating cells as a venue to establish mixed chimerism and tolerance.Pediatr Transplant. 2003 Oct;7(5):348-57. doi: 10.1034/j.1399-3046.2003.00100.x. Pediatr Transplant. 2003. PMID: 14738294 Review.
-
Donor programmed cell death 1 ligand 1 is required for organ transplant tolerance in major histocompatibility complex-mismatched mixed chimeras although programmed cell death 1 ligand 1 and major histocompatibility complex class II are not required for inducing chimerism.Am J Transplant. 2023 Aug;23(8):1116-1129. doi: 10.1016/j.ajt.2023.04.022. Epub 2023 Apr 25. Am J Transplant. 2023. PMID: 37105316
-
Protective conditioning against GVHD and graft rejection after combined organ and hematopoietic cell transplantation.Blood Cells Mol Dis. 2008 Jan-Feb;40(1):48-54. doi: 10.1016/j.bcmd.2007.06.019. Epub 2007 Sep 10. Blood Cells Mol Dis. 2008. PMID: 17827036 Review.
-
Methyl-Guanine-Methyl-Transferase Transgenic Bone Marrow Transplantation Allows N,N-bis(2-chloroethyl)-Nitrosourea Driven Donor Mixed-Chimerism Without Graft-Versus-Host Disease, and With Donor-Specific Allograft Tolerance.Transplantation. 2015 Dec;99(12):2476-84. doi: 10.1097/TP.0000000000000825. Transplantation. 2015. PMID: 26177088
Cited by
-
Micro-chimerism: from evolution to revolution.Semin Immunopathol. 2025 Aug 6;47(1):33. doi: 10.1007/s00281-025-01060-x. Semin Immunopathol. 2025. PMID: 40770572 Free PMC article. No abstract available.
-
Kidney transplants from elderly donors: what we have learned 20 years after the Crystal City consensus criteria meeting.J Nephrol. 2024 Jul;37(6):1449-1461. doi: 10.1007/s40620-024-01888-w. Epub 2024 Mar 6. J Nephrol. 2024. PMID: 38446386 Free PMC article. Review.
-
Targeting Macrophages in Organ Transplantation: A Step Toward Personalized Medicine.Transplantation. 2024 Oct 1;108(10):2045-2056. doi: 10.1097/TP.0000000000004978. Epub 2024 Mar 12. Transplantation. 2024. PMID: 38467591 Review.
-
Animal models for transplant immunology: bridging bench to bedside.Clin Transplant Res. 2024 Dec 31;38(4):354-376. doi: 10.4285/ctr.24.0029. Epub 2024 Sep 5. Clin Transplant Res. 2024. PMID: 39233453 Free PMC article. Review.
-
Initial exploration of a novel fusion protein, IL-4/IL-34/IL-10, which promotes cardiac allograft survival mice through alloregulation.Innate Immun. 2023 Oct;29(7):150-158. doi: 10.1177/17534259231186239. Innate Immun. 2023. PMID: 37800911 Free PMC article.
References
-
- Schnuelle P, Lorenz D, Trede M, van der Woude FJ. Impact of Renal Cadaveric Transplantation on Survival in End-Stage Renal Failure: Evidence for Reduced Mortality Risk Compared With Hemodialysis During Long-Term Follow-Up. J Am Soc Nephrol (1998) 9(11):2135–41. doi: 10.1681/ASN.V9112135 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

