Vascular Stem/Progenitor Cells in Vessel Injury and Repair
- PMID: 35224067
- PMCID: PMC8866648
- DOI: 10.3389/fcvm.2022.845070
Vascular Stem/Progenitor Cells in Vessel Injury and Repair
Abstract
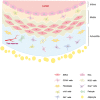
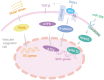
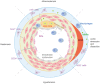
Vascular repair upon vessel injury is essential for the maintenance of arterial homeostasis and function. Stem/progenitor cells were demonstrated to play a crucial role in regeneration and replenishment of damaged vascular cells during vascular repair. Previous studies revealed that myeloid stem/progenitor cells were the main sources of tissue regeneration after vascular injury. However, accumulating evidences from developing lineage tracing studies indicate that various populations of vessel-resident stem/progenitor cells play specific roles in different process of vessel injury and repair. In response to shear stress, inflammation, or other risk factors-induced vascular injury, these vascular stem/progenitor cells can be activated and consequently differentiate into different types of vascular wall cells to participate in vascular repair. In this review, mechanisms that contribute to stem/progenitor cell differentiation and vascular repair are described. Targeting these mechanisms has potential to improve outcome of diseases that are characterized by vascular injury, such as atherosclerosis, hypertension, restenosis, and aortic aneurysm/dissection. Future studies on potential stem cell-based therapy are also highlighted.
Keywords: aortic aneurysm; atherosclerosis; hypertension; restenosis; vascular injury; vascular remodeling; vascular stem cells.
Copyright © 2022 Tao, Cao, Yu and Qu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources

