PreserFlo® MicroShunt: An Overview of This Minimally Invasive Device for Open-Angle Glaucoma
- PMID: 35225971
- PMCID: PMC8883991
- DOI: 10.3390/vision6010012
PreserFlo® MicroShunt: An Overview of This Minimally Invasive Device for Open-Angle Glaucoma
Abstract
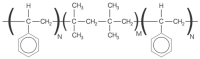
For moderate-to-severe glaucoma, trabeculectomy remains the "gold standard" intraocular pressure (IOP)-lowering treatment; nonetheless, this method requires extensive post-operative maintenance. Microinvasive glaucoma surgery (MIGS) treatments are designed to lessen intra- and post-operative care burden while offering an acceptable IOP decrease for individuals with mild to moderate glaucoma. The PreserFlo® MicroShunt (previously InnFocus MicroShunt) is an 8.5 mm glaucoma drainage device manufactured from poly(styrene-block-isobutylene-block-styrene) (SIBS), an extremely biocompatible and bioinert material. The lumen is narrow enough to prevent hypotony, but big enough to avoid being obstructed by sloughed cells or pigment. The device is implanted ab externo, as a stand-alone procedure or in conjunction with cataract surgery, with intraoperative mitomycin C, and a bleb is produced under the conjunctiva and Tenon's capsule. The MicroShunt was CE-marked in 2012 and designed for primary open-angle glaucoma, the IOP of which remains uncontrolled after maximally tolerated topical treatment. Several clinical trials evaluating the MicroShunt's long-term safety and effectiveness have been conducted, highlighting the effectiveness of the device over time, along with a tolerable safety profile. The present review aims to gather evidence of PreserFlo's effectiveness and safety results almost 10 years after its introduction, and furthermore, to compare it with other MIGS and with the gold-standard trabeculectomy for glaucoma management.
Keywords: InnFocus MicroShunt; MicroShunt; PreserFlo; PreserFlo MicroShunt; SIBS polymer; XEN Gel Stent; glaucoma; glaucoma drainage devices; micro-invasive glaucoma surgery; mitomycin C.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Ab externo implantation of the MicroShunt, a poly (styrene-block-isobutylene-block-styrene) surgical device for the treatment of primary open-angle glaucoma: a review.Eye Vis (Lond). 2019 Nov 15;6:36. doi: 10.1186/s40662-019-0162-1. eCollection 2019. Eye Vis (Lond). 2019. PMID: 31807606 Free PMC article. Review.
-
PreserFlo MicroShunt® exposure: a case series.BMC Ophthalmol. 2021 Jul 10;21(1):273. doi: 10.1186/s12886-021-02032-z. BMC Ophthalmol. 2021. PMID: 34246229 Free PMC article.
-
Safety and Effectiveness of the PRESERFLO® MicroShunt in Primary Open-Angle Glaucoma: Results from a 2-Year Multicenter Study.Ophthalmol Glaucoma. 2022 Mar-Apr;5(2):195-209. doi: 10.1016/j.ogla.2021.07.008. Epub 2021 Jul 28. Ophthalmol Glaucoma. 2022. PMID: 34329772
-
Subconjunctival filtration in evolution: current evidence on MicroShunt implantation for treating patients with glaucoma.Eye Vis (Lond). 2023 Mar 2;10(1):10. doi: 10.1186/s40662-022-00322-1. Eye Vis (Lond). 2023. PMID: 36859515 Free PMC article. Review.
-
The PreserFlo MicroShunt in the Context of Minimally Invasive Glaucoma Surgery: A Narrative Review.Int J Environ Res Public Health. 2023 Feb 7;20(4):2904. doi: 10.3390/ijerph20042904. Int J Environ Res Public Health. 2023. PMID: 36833599 Free PMC article. Review.
Cited by
-
Recanalization of Xen45 gel stent implant occlusion using 10 - 0 nylon suture in refractory glaucoma: a case report.BMC Ophthalmol. 2023 Oct 19;23(1):418. doi: 10.1186/s12886-023-03109-7. BMC Ophthalmol. 2023. PMID: 37858210 Free PMC article.
-
Early Results of PreserFlo MicroShunt Implantation in Complicated Cases: A Single-Center Case Series.Cureus. 2025 Apr 28;17(4):e83116. doi: 10.7759/cureus.83116. eCollection 2025 Apr. Cureus. 2025. PMID: 40438849 Free PMC article.
-
PRESERFLO MicroShunt implantation versus trabeculectomy for primary open-angle glaucoma: a two-year follow-up study.Eye Vis (Lond). 2023 Dec 21;10(1):50. doi: 10.1186/s40662-023-00369-8. Eye Vis (Lond). 2023. PMID: 38124210 Free PMC article.
-
Efficacy and Safety of PreserFlo MicroShunt Implantation and Its Effects on Intraocular Inflammation through Laser Flare Photometry.J Ophthalmol. 2024 Jul 23;2024:2447721. doi: 10.1155/2024/2447721. eCollection 2024. J Ophthalmol. 2024. PMID: 39081855 Free PMC article.
-
The Paul Glaucoma Implant: a systematic review of safety, efficacy, and emerging applications.Graefes Arch Clin Exp Ophthalmol. 2025 May 28. doi: 10.1007/s00417-025-06861-2. Online ahead of print. Graefes Arch Clin Exp Ophthalmol. 2025. PMID: 40434721 Review.
References
-
- Gedde S.J., Feuer W.J., Shi W., Lim K.S., Barton K., Goyal S., Ahmed I.I.K., Brandt J., Primary Tube Versus Trabeculectomy Study G. Treatment Outcomes in the Primary Tube Versus Trabeculectomy Study after 1 Year of Follow-up. Ophthalmology. 2018;125:650–663. doi: 10.1016/j.ophtha.2018.02.003. - DOI - PubMed
-
- Gedde S.J., Schiffman J.C., Feuer W.J., Herndon L.W., Brandt J.D., Budenz D.L., Tube versus Trabeculectomy Study G. Treatment outcomes in the Tube Versus Trabeculectomy (TVT) study after five years of follow-up. Am. J. Ophthalmol. 2012;153:789–803.e2. doi: 10.1016/j.ajo.2011.10.026. - DOI - PMC - PubMed
-
- Gedde S.J., Herndon L.W., Brandt J.D., Budenz D.L., Feuer W.J., Schiffman J.C., Tube Versus Trabeculectomy Study G. Postoperative complications in the Tube Versus Trabeculectomy (TVT) study during five years of follow-up. Am. J. Ophthalmol. 2012;153:804–814.e1. doi: 10.1016/j.ajo.2011.10.024. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

