Ecological and social pressures interfere with homeostatic sleep regulation in the wild
- PMID: 35229719
- PMCID: PMC8887896
- DOI: 10.7554/eLife.73695
Ecological and social pressures interfere with homeostatic sleep regulation in the wild
Abstract
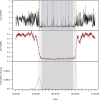
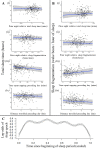
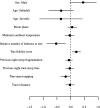
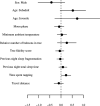
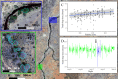
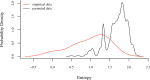
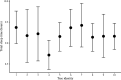
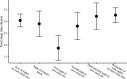
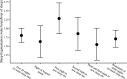
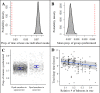
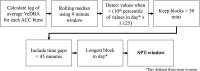
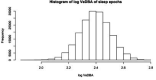
Sleep is fundamental to the health and fitness of all animals. The physiological importance of sleep is underscored by the central role of homeostasis in determining sleep investment - following periods of sleep deprivation, individuals experience longer and more intense sleep bouts. Yet, most sleep research has been conducted in highly controlled settings, removed from evolutionarily relevant contexts that may hinder the maintenance of sleep homeostasis. Using triaxial accelerometry and GPS to track the sleep patterns of a group of wild baboons (Papio anubis), we found that ecological and social pressures indeed interfere with homeostatic sleep regulation. Baboons sacrificed time spent sleeping when in less familiar locations and when sleeping in proximity to more group-mates, regardless of how long they had slept the prior night or how much they had physically exerted themselves the preceding day. Further, they did not appear to compensate for lost sleep via more intense sleep bouts. We found that the collective dynamics characteristic of social animal groups persist into the sleep period, as baboons exhibited synchronized patterns of waking throughout the night, particularly with nearby group-mates. Thus, for animals whose fitness depends critically on avoiding predation and developing social relationships, maintaining sleep homeostasis may be only secondary to remaining vigilant when sleeping in risky habitats and interacting with group-mates during the night. Our results highlight the importance of studying sleep in ecologically relevant contexts, where the adaptive function of sleep patterns directly reflects the complex trade-offs that have guided its evolution.
Keywords: biotelemetry; ecology; evolutionary biology; homeostasis; olive baboon; predation risk; sleep; social behavior.
© 2022, Loftus et al.
Conflict of interest statement
JL, RH, CN, MC No competing interests declared
Figures



Comment in
-
Accelerometer-based analyses of animal sleep patterns.Elife. 2022 Mar 8;11:e77349. doi: 10.7554/eLife.77349. Elife. 2022. PMID: 35258454 Free PMC article.
References
-
- Altmann SA, Altmann J. Baboon Ecology: African Field Research. University of Chicago Press; 1970. - DOI
-
- Amlaner CJ, Phil D, Fuller PM. Basics of Sleep Guide. Sleep Research Society; 2009.
-
- Bäckman J, Andersson A, Pedersen L, Sjöberg S, Tøttrup AP, Alerstam T. Actogram analysis of free-flying migratory birds: new perspectives based on acceleration logging. Journal of Comparative Physiology. A, Neuroethology, Sensory, Neural, and Behavioral Physiology. 2017;203:543–564. doi: 10.1007/s00359-017-1165-9. - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources

