Interaction of background Ca2+ influx, sarcoplasmic reticulum threshold and heart failure in determining propensity for Ca2+ waves in sheep heart
- PMID: 35233776
- PMCID: PMC9310721
- DOI: 10.1113/JP282168
Interaction of background Ca2+ influx, sarcoplasmic reticulum threshold and heart failure in determining propensity for Ca2+ waves in sheep heart
Abstract
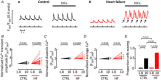
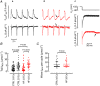
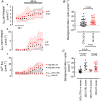
Ventricular arrhythmias can cause death in heart failure (HF). A trigger is the occurrence of Ca2+ waves which activate a Na+ -Ca2+ exchange (NCX) current, leading to delayed after-depolarisations and triggered action potentials. Waves arise when sarcoplasmic reticulum (SR) Ca2+ content reaches a threshold and are commonly induced experimentally by raising external Ca2+ , although the mechanism by which this causes waves is unclear and was the focus of this study. Intracellular Ca2+ was measured in voltage-clamped ventricular myocytes from both control sheep and those subjected to rapid pacing to produce HF. Threshold SR Ca2+ content was determined by applying caffeine (10 mM) following a wave and integrating wave and caffeine-induced NCX currents. Raising external Ca2+ induced waves in a greater proportion of HF cells than control. The associated increase of SR Ca2+ content was smaller in HF due to a lower threshold. Raising external Ca2+ had no effect on total influx via the L-type Ca2+ current, ICa-L , and increased efflux on NCX. Analysis of sarcolemmal fluxes revealed substantial background Ca2+ entry which sustains Ca2+ efflux during waves in the steady state. Wave frequency and background Ca2+ entry were decreased by Gd3+ or the TRPC6 inhibitor BI 749327. These agents also blocked Mn2+ entry. Inhibiting connexin hemi-channels, TRPC1/4/5, L-type channels or NCX had no effect on background entry. In conclusion, raising external Ca2+ induces waves via a background Ca2+ influx through TRPC6 channels. The greater propensity to waves in HF results from increased background entry and decreased threshold SR content. KEY POINTS: Heart failure is a pro-arrhythmic state and arrhythmias are a major cause of death. At the cellular level, Ca2+ waves resulting in delayed after-depolarisations are a key trigger of arrhythmias. Ca2+ waves arise when the sarcoplasmic reticulum (SR) becomes overloaded with Ca2+ . We investigate the mechanism by which raising external Ca2+ causes waves, and how this is modified in heart failure. We demonstrate that a novel sarcolemmal background Ca2+ influx via the TRPC6 channel is responsible for SR Ca2+ overload and Ca2+ waves. The increased propensity for Ca2+ waves in heart failure results from an increase of background influx, and a lower threshold SR content. The results of the present study highlight a novel mechanism by which Ca2+ waves may arise in heart failure, providing a basis for future work and novel therapeutic targets.
Keywords: Ca2+; heart failure; sarcoplasmic reticulum; threshold; waves.
© 2022 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Figures





Comment in
-
Background calcium influx in arrhythmia: lead actor or extra?J Physiol. 2022 Jun;600(11):2545-2546. doi: 10.1113/JP283032. Epub 2022 May 13. J Physiol. 2022. PMID: 35451079 No abstract available.
-
The role of background calcium influx on heart failure-associated calcium waves: what you do not know, you do not recognise.J Physiol. 2022 Jun;600(11):2561-2564. doi: 10.1113/JP283146. Epub 2022 May 11. J Physiol. 2022. PMID: 35476213 No abstract available.
References
-
- Ai X, Curran JW, Shannon TR, Bers DM & Pogwizd SM (2005). Ca2+/calmodulin‐dependent protein kinase modulates cardiac ryanodine receptor phosphorylation and sarcoplasmic reticulum Ca2+ leak in heart failure. Circ Res 97, 1314–1322. - PubMed
-
- Bassani RA, Bassani JW & Bers DM (1995). Relaxation in ferret ventricular myocytes: role of the sarcolemmal Ca ATPase. Pflugers Arch 430, 573–578. - PubMed
-
- Bers DM (2008). Calcium cycling and signaling in cardiac myocytes. Annu Rev Physiol 70, 23–49. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

