Efficacy of covid-19 vaccines in immunocompromised patients: systematic review and meta-analysis
- PMID: 35236664
- PMCID: PMC8889026
- DOI: 10.1136/bmj-2021-068632
Efficacy of covid-19 vaccines in immunocompromised patients: systematic review and meta-analysis
Abstract
Objective: To compare the efficacy of covid-19 vaccines between immunocompromised and immunocompetent people.
Design: Systematic review and meta-analysis.
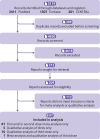
Data sources: PubMed, Embase, Central Register of Controlled Trials, COVID-19 Open Research Dataset Challenge (CORD-19), and WHO covid-19 databases for studies published between 1 December 2020 and 5 November 2021. ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform were searched in November 2021 to identify registered but as yet unpublished or ongoing studies.
Study selection: Prospective observational studies comparing the efficacy of covid-19 vaccination in immunocompromised and immunocompetent participants.
Methods: A frequentist random effects meta-analysis was used to separately pool relative and absolute risks of seroconversion after the first and second doses of a covid-19 vaccine. Systematic review without meta-analysis of SARS-CoV-2 antibody titre levels was performed after first, second, and third vaccine doses and the seroconversion rate after a third dose. Risk of bias and certainty of evidence were assessed.
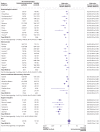
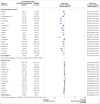
Results: 82 studies were included in the meta-analysis. Of these studies, 77 (94%) used mRNA vaccines, 16 (20%) viral vector vaccines, and 4 (5%) inactivated whole virus vaccines. 63 studies were assessed to be at low risk of bias and 19 at moderate risk of bias. After one vaccine dose, seroconversion was about half as likely in patients with haematological cancers (risk ratio 0.40, 95% confidence interval 0.32 to 0.50, I2=80%; absolute risk 0.29, 95% confidence interval 0.20 to 0.40, I2=89%), immune mediated inflammatory disorders (0.53, 0.39 to 0.71, I2=89%; 0.29, 0.11 to 0.58, I2=97%), and solid cancers (0.55, 0.46 to 0.65, I2=78%; 0.44, 0.36 to 0.53, I2=84%) compared with immunocompetent controls, whereas organ transplant recipients were 16 times less likely to seroconvert (0.06, 0.04 to 0.09, I2=0%; 0.06, 0.04 to 0.08, I2=0%). After a second dose, seroconversion remained least likely in transplant recipients (0.39, 0.32 to 0.46, I2=92%; 0.35, 0.26 to 0.46), with only a third achieving seroconversion. Seroconversion was increasingly likely in patients with haematological cancers (0.63, 0.57 to 0.69, I2=88%; 0.62, 0.54 to 0.70, I2=90%), immune mediated inflammatory disorders (0.75, 0.69 to 0.82, I2=92%; 0.77, 0.66 to 0.85, I2=93%), and solid cancers (0.90, 0.88 to 0.93, I2=51%; 0.89, 0.86 to 0.91, I2=49%). Seroconversion was similar between people with HIV and immunocompetent controls (1.00, 0.98 to 1.01, I2=0%; 0.97, 0.83 to 1.00, I2=89%). Systematic review of 11 studies showed that a third dose of a covid-19 mRNA vaccine was associated with seroconversion among vaccine non-responders with solid cancers, haematological cancers, and immune mediated inflammatory disorders, although response was variable in transplant recipients and inadequately studied in people with HIV and those receiving non-mRNA vaccines.
Conclusion: Seroconversion rates after covid-19 vaccination were significantly lower in immunocompromised patients, especially organ transplant recipients. A second dose was associated with consistently improved seroconversion across all patient groups, albeit at a lower magnitude for organ transplant recipients. Targeted interventions for immunocompromised patients, including a third (booster) dose, should be performed.
Systematic review registration: PROSPERO CRD42021272088.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: no support from any organisation for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
Figures

References
-
- Voysey M, Clemens SAC, Madhi SA, et al. Oxford COVID Vaccine Trial Group . Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021;397:99-111. 10.1016/S0140-6736(20)32661-1 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
