Therapeutic safety and efficacy of triple-immunosuppressants versus dual-immunosuppressants in severe-to-critical COVID-19: a prospective cohort study in Bangladesh
- PMID: 35238257
- PMCID: PMC8903771
- DOI: 10.1080/07853890.2022.2039958
Therapeutic safety and efficacy of triple-immunosuppressants versus dual-immunosuppressants in severe-to-critical COVID-19: a prospective cohort study in Bangladesh
Abstract
Background: Hyperinflammation-induced respiratory failure is a leading cause of mortality in COVID-19 infection. Immunosuppressants such as, Baricitinib and interleukin inhibitors are the drug-of-choice to suppress cytokine storm in COVID-19. Here, we compared the therapeutic safety and efficacy of triple-immunosuppressants with dual-immunosuppressants in patients with severe-to-critical COVID-19.
Methods: This study was conducted on 103 confirmed COVID-19 patients. Of 103 patients, 49 (N) and 54 (N) patients received dual-immunosuppressants (baricitinib plus two doses of secukinumab) and triple immunosuppressants (baricitinib plus single dose of tocilizumab and secukinumab) in group A and group B, respectively. Groups were compared in terms of clinical outcome, critical support-requirement, survival, re-hospitalisation, and adverse events (AEs).
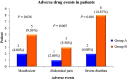
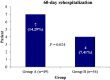
Results: Patients in group B achieved normal blood oxygen saturation level (SpO2) earlier than the patients of group A [4 day (IQR: 3-12) vs 5 day (IQR: 5-14), p < .05]. The requirement of intensive care unit (ICU) and mechanical ventilation (MV) support was less in group B than group A [16.7%/28.6%, 11.1%/18.4%, respectively p < .05]]. The incidence of COVID-19 acute respiratory distress syndrome and 60-day all cause mortality was reduced in group B compared to group A [0.43 (0.19-0.98), p < .05; 0.35 (0.08-1.44), p > .05]. The 60-day re-hospitalisation rate was two-fold high in group A than group B (p = .024). Immunosuppressant-associated adverse events and secondary bacterial/fungal infections were relative high in patients of group B.
Conclusions: Triple-immunosuppressants in severe-to-critical COVID-19 infection exhibited better clinical outcome; reduced ICU and MV requirement; shorter hospital stay with deceased 60-day all cause mortality and re-hospitalisation compared to dual-immunosuppressants.
Keywords: COVID-19; baricitinib; cytokine storm; immunosuppressant; secukinumab; tocilizumab.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures

Similar articles
-
A Phase I/II Clinical Trial to evaluate the efficacy of baricitinib to prevent respiratory insufficiency progression in onco-hematological patients affected with COVID19: A structured summary of a study protocol for a randomised controlled trial.Trials. 2021 Feb 5;22(1):116. doi: 10.1186/s13063-021-05072-4. Trials. 2021. PMID: 33546739 Free PMC article.
-
Impact of high dose of baricitinib in severe COVID-19 pneumonia: a prospective cohort study in Bangladesh.BMC Infect Dis. 2021 May 7;21(1):427. doi: 10.1186/s12879-021-06119-2. BMC Infect Dis. 2021. PMID: 33962573 Free PMC article. Clinical Trial.
-
Safety and Efficacy of Imatinib for Hospitalized Adults with COVID-19: A structured summary of a study protocol for a randomised controlled trial.Trials. 2020 Oct 28;21(1):897. doi: 10.1186/s13063-020-04819-9. Trials. 2020. PMID: 33115543 Free PMC article.
-
Clinical efficacy and adverse events of baricitinib treatment for coronavirus disease-2019 (COVID-19): A systematic review and meta-analysis.J Med Virol. 2022 Apr;94(4):1523-1534. doi: 10.1002/jmv.27482. Epub 2021 Dec 13. J Med Virol. 2022. PMID: 34846766 Free PMC article.
-
Recommendations for coronavirus infection in rheumatic diseases treated with biologic therapy.J Autoimmun. 2020 May;109:102442. doi: 10.1016/j.jaut.2020.102442. Epub 2020 Apr 2. J Autoimmun. 2020. PMID: 32253068 Free PMC article. Review.
References
-
- World Health Organization . WHO Coronavirus Disease (COVID-19). 2021. https://www.who.int/emergencies/diseases/novel-coronavirus-2019. - PubMed
-
- World Health Organization . WHO coronavirus (COVID-19) dashboard. 2021. https://covid19.who.int/.
-
- World Health Organization . WHO Bangladesh COVID-19 morbidity and mortality weekly update. 2021. https://cdn.who.int/media/docs/default-source/searo/bangladesh/covid-19-....
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
