Finerenone in patients with chronic kidney disease and type 2 diabetes with and without heart failure: a prespecified subgroup analysis of the FIDELIO-DKD trial
- PMID: 35239204
- PMCID: PMC9541504
- DOI: 10.1002/ejhf.2469
Finerenone in patients with chronic kidney disease and type 2 diabetes with and without heart failure: a prespecified subgroup analysis of the FIDELIO-DKD trial
Abstract
Aims: This prespecified analysis of the FIDELIO-DKD trial compared the effects of finerenone, a selective non-steroidal mineralocorticoid receptor antagonist, on cardiorenal outcomes in patients with chronic kidney disease (CKD) and type 2 diabetes (T2D) by history of heart failure (HF).
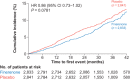
Methods and results: Patients with T2D and CKD (urine albumin-to-creatinine ratio ≥30-5000 mg/g and estimated glomerular filtration rate [eGFR] ≥25-<75 ml/min/1.73 m2 ), without symptomatic HF with reduced ejection fraction (New York Heart Association II-IV) and treated with optimized renin-angiotensin system blockade were randomized to finerenone or placebo. The composite cardiovascular (CV) outcome (CV death, non-fatal myocardial infarction, non-fatal stroke, or hospitalization for HF) and composite kidney outcome (kidney failure, sustained ≥40% decrease in eGFR from baseline, or renal death) were analysed by investigator-reported medical history of HF. Of 5674 patients, 436 (7.7%) had a history of HF. Over a median follow-up of 2.6 years, the effect of finerenone compared with placebo on the composite CV outcome was consistent in patients with and without a history of HF (hazard ratio [HR] 0.73, 95% confidence interval [CI] 0.50-1.06 and HR 0.90, 95% CI 0.77-1.04, respectively; interaction p = 0.33). The effect of finerenone on the composite kidney outcome did not differ by history of HF (HR 0.79, 95% CI 0.52-1.20 and HR 0.83, 95% CI 0.73-0.94, respectively; interaction p = 0.83).
Conclusion: In FIDELIO-DKD, finerenone improved cardiorenal outcome in patients with CKD and T2D irrespective of baseline HF history.
Keywords: Aldosterone; Chronic kidney disease; Diabetes; Finerenone; Heart failure; Mineralocorticoid receptor antagonists.
© 2022 The Authors. European Journal of Heart Failure published by John Wiley & Sons Ltd on behalf of European Society of Cardiology.
Figures


References
-
- Seferovic PM, Petrie MC, Filippatos GS, Anker SD, Rosano G, Bauersachs J, et al. Type 2 diabetes mellitus and heart failure: a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2018;20:853–72. - PubMed
-
- Filippatos G, Farmakis D, Parissis J. Renal dysfunction and heart failure: things are seldom what they seem. Eur Heart J. 2014;35:416–8. - PubMed
-
- Anker SD, Butler J, Filippatos G, Shahzeb Khan M, Ferreira JP, Bocchi E, et al.; EMPEROR‐Preserved Trial Committees and Investigators . Baseline characteristics of patients with heart failure with preserved ejection fraction in the EMPEROR‐Preserved trial. Eur J Heart Fail. 2020;22:2383–92. - PubMed
-
- Solomon SD, Rizkala AR, Lefkowitz MP, Shi VC, Gong J, Anavekar N, et al. Baseline characteristics of patients with heart failure and preserved ejection fraction in the PARAGON‐HF trial. Circ Heart Fail. 2018;11:e004962. - PubMed
-
- Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129–200. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

