Mathematical modeling of therapeutic neural stem cell migration in mouse brain with and without brain tumors
- PMID: 35240798
- PMCID: PMC8958926
- DOI: 10.3934/mbe.2022119
Mathematical modeling of therapeutic neural stem cell migration in mouse brain with and without brain tumors
Abstract
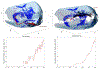
Neural stem cells (NSCs) offer a potential solution to treating brain tumors. This is because NSCs can circumvent the blood-brain barrier and migrate to areas of damage in the central nervous system, including tumors, stroke, and wound injuries. However, for successful clinical application of NSC treatment, a sufficient number of viable cells must reach the diseased or damaged area(s) in the brain, and evidence suggests that it may be affected by the paths the NSCs take through the brain, as well as the locations of tumors. To study the NSC migration in brain, we develop a mathematical model of therapeutic NSC migration towards brain tumor, that provides a low cost platform to investigate NSC treatment efficacy. Our model is an extension of the model developed in Rockne et al. (PLoS ONE 13, e0199967, 2018) that considers NSC migration in non-tumor bearing naive mouse brain. Here we modify the model in Rockne et al. in three ways: (i) we consider three-dimensional mouse brain geometry, (ii) we add chemotaxis to model the tumor-tropic nature of NSCs into tumor sites, and (iii) we model stochasticity of migration speed and chemosensitivity. The proposed model is used to study migration patterns of NSCs to sites of tumors for different injection strategies, in particular, intranasal and intracerebral delivery. We observe that intracerebral injection results in more NSCs arriving at the tumor site(s), but the relative fraction of NSCs depends on the location of injection relative to the target site(s). On the other hand, intranasal injection results in fewer NSCs at the tumor site, but yields a more even distribution of NSCs within and around the target tumor site(s).
Keywords: LM-NSC008; agent based modeling; glioma; intranasal drug administration; mathematical oncology; neural stem cell therapy.
Conflict of interest statement
Conflict of interest
The authors declare that there is no conflict of interest.
Figures



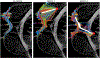





References
-
- Gutova M, Cheng JP, Adhikarla V, Tsaturyan L, Barish ME, Rockne RC, et al. , Intranasally administered L-Myc-immortalized human neural stem cells migrate to primary and distal sites of damage after cortical impact and enhance spatial learning, Stem Cells Int., 2021 (2021), 1–11. 10.1155/2021/5549381. - DOI - PMC - PubMed
