PROPHYlactic Implantation of BIOlogic Mesh in peritonitis (PROPHYBIOM): a prospective multicentric randomized controlled trial
- PMID: 35246206
- PMCID: PMC8895107
- DOI: 10.1186/s13063-022-06103-4
PROPHYlactic Implantation of BIOlogic Mesh in peritonitis (PROPHYBIOM): a prospective multicentric randomized controlled trial
Abstract
Background: Patients undergoing emergency surgery for peritonitis are at increased risk of abdominal wall-related complications. In patients with peritonitis, the risk of incisional hernia (IH) is extremely elevated. The evaluation of quality of life of patients with incisional hernia showed lower mean scores on physical components of health-related quality of life and body image. Furthermore, the arise of a post-operative abdominal wall complication (i.e., wound dehiscence, evisceration and IH) greatly increases morbidity and mortality rates and prolongs the hospitalization.
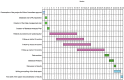
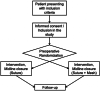
Methods: The present study aims to evaluate the efficacy of the use of a swine dermal collagen prosthesis implanted preperitoneally as a prophylactic procedure in urgency/emergency setting against abdominal wall complications in patients operated with contaminated/infected field in peritonitis. The sample size was defined in 90 patients divided in two arms (prosthesis positioning versus normal wall abdominal closure). The follow-up will be performed at 3, 6, and 12 months after surgery. The percentage of incisional hernias, wound infections, and adverse events will be investigated by physical examination and ultrasound.
Discussion: The objective is to evaluate the possibility to reduce the incisional hernia rate in patients undergoing urgent/emergent laparotomy in contaminated/infected field with peritonitis by using swine dermal collagen prosthesis preperitoneal positioning as a prophylactic procedure.
Trial registration: ClinicalTrials.gov NCT04681326. Registered (retrospectively after first patient recruited) on 23 December 2020.
Keywords: Abdominal sonography; Acute abdomen; Biological mesh; Emergency surgery; Incisional hernia; Laparotomy; Peritonitis; Randomized trial; Swine dermal collagen prosthesis.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Moussavian MR, et al. Long term follow up for incisional hernia after severe secondary peritonitisincidence and risk factors. Am J Surg. 2010; 10.1016/j.amjsurg.2009.08.043. - PubMed
-
- Sørensen LT, Hemmingsen UB, Kirkeby LT, Kallehave F, Jørgensen LN. Smoking is a risk factor for incisional hernia. Arch Surg. 2005; 10.1001/archsurg.140.2.119. - PubMed
-
- Kurmann A, Beldi G, Vorburger SA, Seiler CA, Candinas D. Laparoscopic incisional hernia repair is feasible and safe after liver transplantation. Surg Endosc. 2010; 10.1007/s00464-009-0799-2. - PubMed
-
- van Ramshorst GH, Eker HH, van der Voet JA, Jeekel J, Lange JF. Long-term outcome study in patients with abdominal wound dehiscence: a comparative study on quality of life, body image, and incisional hernia. J Gastrointest Surg. 2013; 10.1007/s11605-013-2233-2. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical