Stress vulnerability shapes disruption of motor cortical neuroplasticity
- PMID: 35246507
- PMCID: PMC8897461
- DOI: 10.1038/s41398-022-01855-8
Stress vulnerability shapes disruption of motor cortical neuroplasticity
Abstract
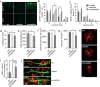
Chronic stress is a major cause of neuropsychiatric conditions such as depression. Stress vulnerability varies individually in mice and humans, measured by behavioral changes. In contrast to affective symptoms, motor retardation as a consequence of stress is not well understood. We repeatedly imaged dendritic spines of the motor cortex in Thy1-GFP M mice before and after chronic social defeat stress. Susceptible and resilient phenotypes were discriminated by symptom load and their motor learning abilities were assessed by a gross and fine motor task. Stress phenotypes presented individual short- and long-term changes in the hypothalamic-pituitary-adrenal axis as well as distinct patterns of altered motor learning. Importantly, stress was generally accompanied by a marked reduction of spine density in the motor cortex and spine dynamics depended on the stress phenotype. We found astrogliosis and altered microglia morphology along with increased microglia-neuron interaction in the motor cortex of susceptible mice. In cerebrospinal fluid, proteomic fingerprints link the behavioral changes and structural alterations in the brain to neurodegenerative disorders and dysregulated synaptic homeostasis. Our work emphasizes the importance of synaptic integrity and the risk of neurodegeneration within depression as a threat to brain health.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

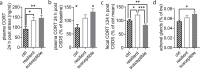


References
-
- Stelzhammer V, Ozcan S, Gottschalk MG, Steeb H, Hodes GE, Guest PC, et al. Central and peripheral changes underlying susceptibility and resistance to social defeat stress—a proteomic profiling study. Diagnostics Neuropsychiatry. 2015;1:1–7. doi: 10.1016/j.dineu.2015.08.001. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

